
Topics in Photographic Preservation 1993, Volume 5, Article 17 (pp. 153-164)
Two wedding photographs mounted onto a sheet of paper and wrapped onto a metal plate were sealed in a plastic coating in Cuba sometime at the beginning of this century. The paper component of this artifact had suffered severe deterioration. Testing indicated the coating to be a cellulose acetate material. The several elements of the collage were separated, cleaned, repaired and re-assembled.
A client brought a bag filled with a mounted photo-collage and various fragments into our laboratory one day in the summer of 1991 (see Figure 1). The object was a composite structure of a metal base covered with a wood pulp paper support with a printed design laminated onto the pulp core. Into this paper cover two oval holes had been cut with a die allowing for the insertion of two photographic images. The edges of the photographs were outlined with a gold leaf paint and the surface of the rose-patterned design laminate paper mount was also speckled with gold. This paper sheet was then forced onto and around the metal base which is the shape of a hubcap. The whole unit was then covered in a plastic coating which gave a rich luster to the image (see figure 2 for an exploded diagram).

Figure 1
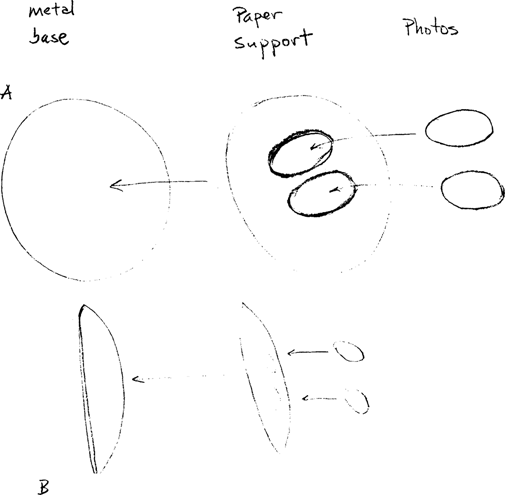
Figure 2
The effect of the coating and the gold speckles must have been quite attractive when the object was first created. When we saw this object in our laboratory, however, the photographs were dark and the paper was very brittle, impossible to manipulate without cracking or flaking. I have only seen one other object of this type before and it was not cracked or torn, but the image also appeared somewhat obscured by a darkened coating. The owner at first wanted to know if the photographs could be removed without damage, but had lost hope for any kind of restoration of the collage. I assumed that the rust on the metal base did not indicate water damage and that the deterioration of the paper support had resulted in both the aging of the coating and the pulp of the paper. Our chemical tests showed that the coating was soluble in acetone, ethyl acetate and ethanol, and partly soluble in amyl acetate, diethylbenzene(softens) and xylene(becomes sticky), it was ignited at low heat before softening(figure 3). Testing was negative for nitrate with test for ammonia using magnesium sulfate and camphor and also negative for nitrogen of NO3 with FeSO4(1) Janowski's Test for nitrate was also negative(2). Testing for acetate was positive using 18M sulfuric acid adding ethyl alcohol, mixing and heating in a boiling water bath(l). The paper support had a pH of less than 4. The coating was severely abraded in addition to its obvious cracking, tearing and resultant losses. The most effective solvent system on samples of the coating was a mixture of acetone and ethyl acetate 1:1 and then 10% addition of amyl acetate. Although the use of these solvents was varied in the process of removal of the coating and the segments of the artifact assemblage. This may have been due to differential aging of the coating.

Figure 3
Examination of the photographic image under 30 power magnification indicated a gelatin silver print structure(3), although this was made difficult by the degree of darkening of the coating film. Microscopic examination also lead to the possibility that the images had been tinted or colored in some way. Treatment would depend on the stability of these pigments or dyes in any solvent system. Since the structure of the photographic paper used in these images is less porous than those reported in other studies(4), we were unsure both of the effects of treatment on the stability of the pigments and the effects of the pigments on the photographic image. Our tests indicated, fortunately, that the solvents proposed in the treatment would have little if any adverse effect on the pigments.
It was apparent that the surface coating was both obscuring the fine photographic qualities of the photograph, in addition to darkening the image, and that its increasing embrittlement was contributing to the overall damage the mount had suffered. These conditions argued strongly for removal of the coating.
The collage fragments were removed from the metal base mechanically after softening the edges in acetone(Figure 4). The photographic images were tested with the solvents which dissolved the coating to determine any adverse effects. This was executed with a thin brush and a cotton swab under 30 power magnification. Examination of the verso of the collage indicated that application of solvent along the edges of the attachment of the photos and the paper mount might free the photographs as the photographs appeared to be set in without a continuous backing. Separation of the photographs was accomplished in this way using micropipet delivery of solvent controlled with a cotton swab. Removal of the coating from the photographs was begun using swabs alternating acetone and ethyl acetate. The coating was removed from the paper support using a bath of the same solvents in a 1:1 ratio. The resulting cleaned major fragments are shown in Figure 5. we were aware that some printing inks contained shellac at the time of the fabrication of this object(5) and that some swelling might occur in the patterned design during treatment. No swelling or other signs of change could be noted in tests and none was observed during treatment.
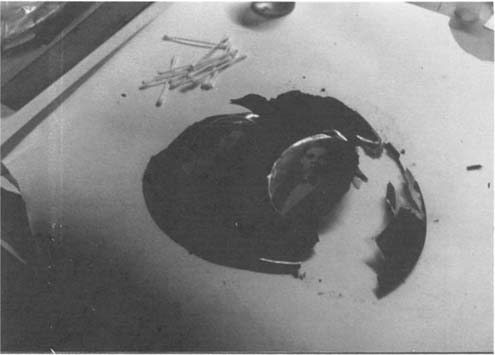
Figure 4

Figure 5
Once cleaned, the possibility of toned or hand-painted coloring was proven with color in the faces and background. The images appeared in fairly good shape and initial appraisal of surface damage due to abrasion were only born out on one(see right image in Figure 6). The remaining fragments of the mount were de-acidified in a bath of 2% Ca(OH)2 and washed in de-ionized water before and after de-acidification. The mount fragments and the photographs were assembled(Figure 7) and then mounted onto a sheet of Japanese Sekishu tissue(Figures 8 & 9). Areas of loss were inpainted with watercolors. The base was cleaned using isopropanol(70% grade) and then swabbed over with a 1% solution of oxalic acid in water(6), this reduced the rust areas and the oxalic acid acted as a chelating agent as well(7). The base was then coated with B-66 in a spray application (8) and a hollow circle of acid-free corrugated board was cut and mounted onto the base using a 5% solution of B-72 in toluene(9) as an adhesive(see Figure 10). The re-constructed photocollage could then be attached to a circular back mat with Japanese paper hinges and wheat starch paste. This could be placed on the top of the corrugated board. The owner could then have a circular frame made for the piece. We recommended a circular top mat be made of acid-free mat board.

Figure 6

Figure 7

Figure 8
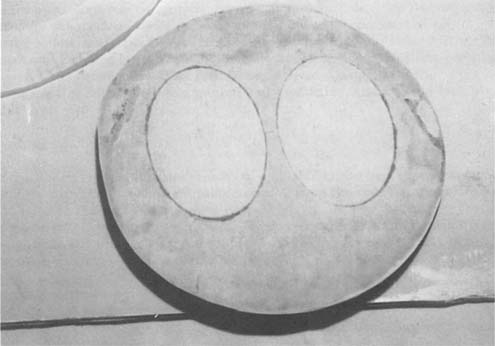
Figure 9
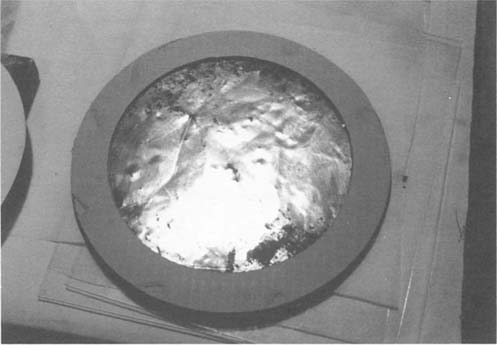
Figure 10
The described treatment achieved the original goal of saving the photographic images and was also able to retain some of the original form of the artifact they were a part. The ethical question here is whether the new reconstructed assemblage still carries the authentic character of the original. The differences are both subtle and striking: the new format is flat and not formed onto the metal base. This is a change in the intent of construction which the craftsman fashioned. The new format appears without the glossy coating of the original. This was the decision of the owner and also our recommendation that although a coating could be applied which would simulate that of the original, we felt it was best not to do so. I feel that these decisions are in concert with the discussions presented at the 1981 3rd International Restorer Seminar on Problems of Completion, Ethics and Scientific Investigation in Restoration. Concern over the retention and alteration of original photographic enclosures is an important current issue (4,10,11) and deserves further discussion.
We would be very interested in any information on these photocollages, especially background on their history and manufacture. We would also like to hear from other conservators who have treated similar objects. Perhaps an expanded article could be fashioned by several treatment approaches that could provide more perspective.
1. Sorum, C.H., Introduction to Semimicro Qualitative Analysis, Second Edition, Prentice Hall, Englewood Cliffs, 1953. The acetate test is a rather subjective procedure but as Hildebrand & Powell (Principles of Chemistry, 1964) note, this part of the “smell-o-metric” bank of methods has had wide practical application.
2. Benedetti-Pichler, A.A., Identification of Materials: Via Physical Properties, Chemical Tests and Microscopy, Springer-Verlag, Wien, 1965.
3. Gill, Arthur T., “Photographic processes, a glossary and a chart for recognition”, Museum Association Information Sheet, Museum Association, London, 1978: 1–10; Hendricks, Klaus B., “The Conservation of Photographic Materials”, Picturescope, Spring, 1982: 4–11. Rempel, Siefgried, “The Care of Black and White Photographic Collections: Identification of Processes”, Technical Bulletin 6, Canadian Conservation Institute, Nov. 1979; Keefe, Jr., Laurence E. & Inch, Dennis, Life of a Photograph, Focal Press, N.Y., 1984.
4. Hendriks, Klaus B. & Dobrusskin, Sebastian, “The Conservation of Painted Photographs”, 9th Triennial Meeting of ICOM Committee for Conservation, Dresden, 1990: 249–254.
5. Tuttle, D. E. & Hoagland, Stewart, “Aniline”, Print, v. 7, #5,Nov. 1952: 1–8.
6. Plenderleith, H.J. & Werner, A.E.A., The Conservation of Antiquities and Works of Art, 2nd Ed., Oxford U. Press, London, 1971. They recommend a 9% solution, but 1% is quite effective in my experience and, in light of the toxicity of this agent, prudent.
7. Burgess, Helen, “The use of chelating agents in conservation treatments”, The Paper Conservator, v. 15, 1991: 36–44 and also. Richey, W.D., “Chelating agents: a review”, Preprints of the Contributions to the Stockholm Congress: Conservation in Archaeology and the Applied Arts, IIC, 1977: 229–234.
8. B-66 is the acryloid thermoplastic resin component in many of the Krylon Crystal Clear spray products according to Krylon Scientist, John Anderson (Personal Communication, 1993). Formulation of Krylon products seem to have varied over the years, for example, Debra Daly in her An Investigation into the Use of Several Substances for Works of Art in Pastel, Master's Thesis, Queen's University Canada, 1978, states that Krylon was B-72 according to a Rohm & Haas 1961 Reporter (v. 19, #2) notice. But Rohm & Haas sold components to Borden for separate manufacturing in the intervening years and Rohm & Haas Scientist, Teresa Sims told me that records were not available to tell when the change to B-66 occurred. Rohm & Haas Scientist Paul Pierson noted that Borden sold Krylon in the 1980's and that much of the data on Krylon formulations were not at Rohm & Haas or Borden but that he couldn't recall when B-72 had been used, if ever.
9. Koob, Stephen P., “The use of Paraloid B-72-as an adhesive: its application for archaeological ceramics and other materials”, Studies in Conservation, 31, 1986: 7–14.
10. Donnithorne, Alan, “The conservation of historical photographs at the British Museum”, The Paper Conservator, v. 12, 1988: 72–9.
11. Moor, Ian & Moor, Angela, “Physical conservation and restoration of photographs”, The Paper Conservator, v. 12, 1988: 86–92.