Topics in Photographic Preservation 1993, Volume 5, Article 13 (pp. 123-137)
In the past few years more and more films in archives, fine art collections and personal collections have succumbed to acetate deterioration. All acetate films—8mm, 16mm, 35mm and sheet film, x-ray, aerial, micro-, cinema, amateur and professional films—are susceptible to this problem. Until now, advice has been limited to vague statements about keeping film cool and dry. It has only been within the last five years that quantitative data has been available on the effects of storage conditions on the process of deterioration. The IPI Storage Guide for Acetate Film is a quantitative tool for understanding how temperature and humidity affect the rate of film base degradation.
Acetate deterioration or “vinegar syndrome”, as it is sometimes called, is characterized by an increase in acidity in the plastic film base. This degradation is an “autocatalytic” process. A catalyst is simply a substance that is not consumed in a reaction, but that makes the reaction much faster. Probably the best example of a catalytic reaction is the hand warmer sold in grocery stores. The plastic wrapper is opened and the inner bag is shaken well. The bag contains three basic materials: sawdust to distribute and hold the heat, iron filings, and a catalyst. The catalyst accelerates the rusting of the iron filings in air to such an extent that the bag gets hot. In the case of the cellulose acetate, acid is the catalyst. The deterioration process is autocatalytic because the process generates its own catalyst. One acid unit goes into the reaction and two come out. Those two acids go into two reactions and four come out resulting in a slow initial deterioration rate followed by a rapid increase in the later stages. Fortunately, the autocatalytic behavior is influenced by the storage circumstances. Materials kept under good conditions in the early part of their lives tend to last longer. Also, films in more open packages generally degrade slower than tightly sealed films because the acetic acid catalyst is allowed to escape.
In the advanced stages of degradation, the “free acidity”, a measure of the amount of acid in the plastic support, is above 5.0 while in fresh film, these acid levels are between 0.05 and 0.2. Physically, the film at this stage has shrunk 10% to 15%, has channelled and has become very brittle. In addition, the acidic by-products of degradation may cause acid-catalyzed hydrolysis of gelatin binders (causing them to soften) and hydrolysis of the yellow dye in chromogenic films (causing fading).
The data used in the Guide was produced in an NEH-funded study at the Image Permanence Institute between 1988 and 1990. Approximately 15,000 data points from this study were used to make the Guide. Tests were performed at 20%, 50%, 60% and 80% RH to determine the temperature dependence of acetate deterioration to a specified end point at these humidities. Mathematical modelling allowed predictions of deterioration rate to be made from the data for any temperature and any humidity between 20% and 80% RH. The Guide is based on this model.
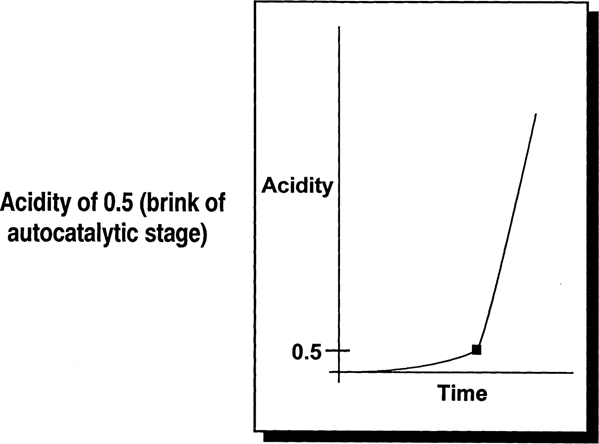
The endpoint selected was 0.5 acidity, a point that is just on the edge of the rapid autocatalytic build-up of acidity. See Fig. 1. At this point, film will have a vinegar odor but will still be usable. Channeling and buckling will not have occurred yet. Film even at the 0.5 acidity level can be preserved by improved storage.

Fig. 1: Basis for Predictions in the IPI Guide
The experimental constraints on the data from which the Guide is made requires that we assume that the storage conditions under consideration are at constant temperature and humidity and that the film is stored in a tightly sealed package. Such assumptions are not unrealistic since motion picture film in closed cans and sheet film tightly packed in drawers in an air-conditioned room would meet them.
With this quantitative measure of how the rate of deterioration is affected by storage conditions, a new term is needed to describe this concept. “Climate Index” or “CI” is the term that IPI has defined as the predicted time in years for a specified change to occur if material is kept under constant environmental conditions. More specifically for acetate film, it is the time in years for fresh film to reach 0.5 free acidity under constant environmental conditions. It is important to note that CI is only an approximate ball-park figure generated from accelerated aging data. Orders of magnitude and relative values are much more accurate than absolute predictions. Also, the higher the predicted CI value, the less certainty there is. It should not be expected that film stored under particular conditions will last exactly as long as the CI. These CI values are like the expiration dates on milk. Some cartons will go bad before the date on the top, while others may last weeks beyond the date.
The Guide itself consists of four tools: a Time Contour, a “Time Out of Storage” Table, a Wheel and an information booklet (Fig. 2). The Time Contour is a contour plot providing a general overview of film life expectancy across a wide range of temperatures and humidities (Fig. 3). Outdoor people will recognize the Time Contour as being similar to a topographic map. In this case “latitude” and “longitude” are the axis of temperature and humidity while the lines of contour are CI values (in years) rather than elevations above sea level. The CI changes at an increasing rate as temperature and humidity decrease. Thus a one-degree change or a one-percent RH change at high temperature and humidity has a much smaller effect on the CI than either a one-degree change or a one-percent RH change at low temperature and humidity.
Fig. 2: The Guide
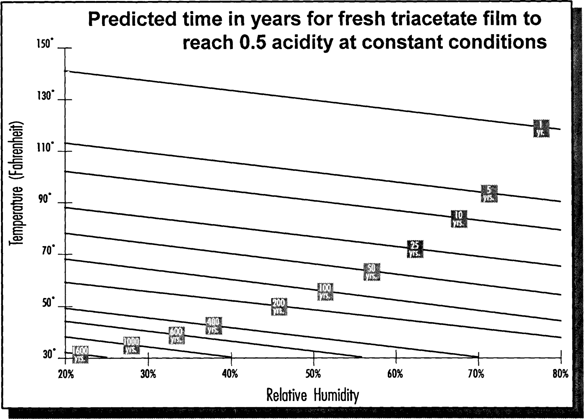
Fig. 3: Time Contours for Vinegar Syndrome
The “Time Out of Storage” Table was the brain child of Mark McCormick-Goodhart at the Conservation Analytical Lab at Smithsonian. He applied the same principles to relative dye fading data from Kodak's publication F-40, The Conservation of Photographs and presented it at AIC in 1992. The Table (partly shown in Fig. 4) has been created from the same data as the contour plot. The general idea behind the Table is that deterioration will occur largely while the film is out of a good storage area. The Table defines an “outside” condition of 75°F/60% RH. The “vault condition” is given by temperature/humidity pairs in the left-hand columns of the Table. Along the top of the Table, columns of “average days out of storage per year” are defined. The Table thus provides CI values for film that is kept in a vault at conditions defined in the left-hand columns that is taken out of storage and into the 75°F/60% RH “outer room” for an average of X days per year. A collection that is on display for three weeks every 20 years is thus out of storage an average of roughly one day per year. Additional days may be added for vault repairs and maintenance. Ninety and 120 days out of storage have been added for teaching staff who may take slides out of storage one quarter or one semester per year. The zero days out of storage column provides CI values for film that is left in the vault and never touched. These figures are the same as are shown on the Wheel and Time Contour. What is important about the Table is that it shows how rapidly the benefits of cold storage are lost as materials are taken out for longer and longer periods. A cold storage vault at −15° F/40% RH for materials that will be kept out one semester per year has no advantage over a cheaper vault at 40° F/40% RH. (Fig. 4)
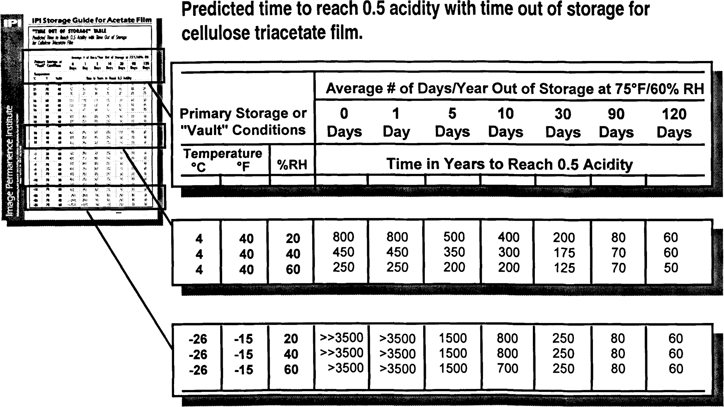
Fig. 4: “Time Out of Storage” Table
The Wheel contains the same data as the Time Contour, arranged for quick estimates of film life expectancy at specific temperature and humidity conditions. Side One of the Wheel is handy for exploring “what if” scenarios or for comparing one storage area with another. Side Two contains similar information for film that is currently on the brink of destruction.
As shown in Fig. 5, fresh film kept at 70° F/50% RH has a CI of only 40 years. Clearly, room conditions are not acceptable for long-term keeping of acetate film. Cool storage of 40° F/50% RH, as shown in Fig. 6, will increase the 40 years to about 350 years. Colder storage, ten degrees lower at 30° F, will further increase the CI by a factor of two (Fig. 7).
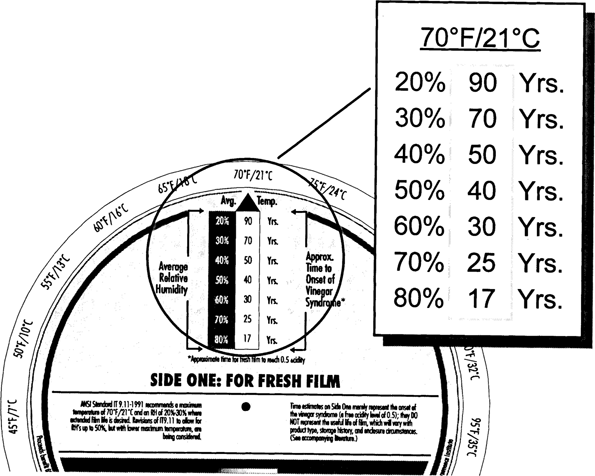
Fig. 5: Climate Index for Fresh Film at 70°F
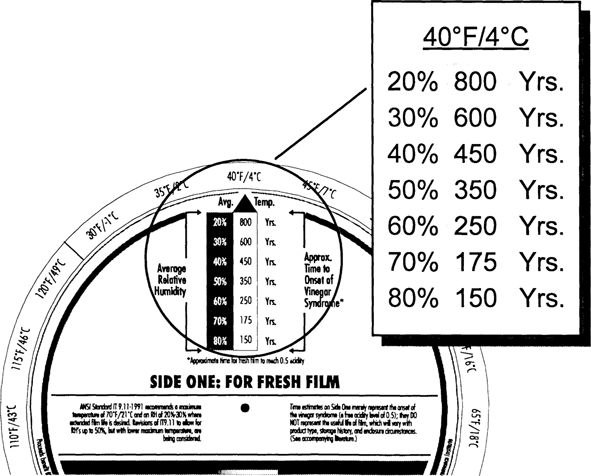
Fig. 6: Climate Index for Fresh Film at 40°F
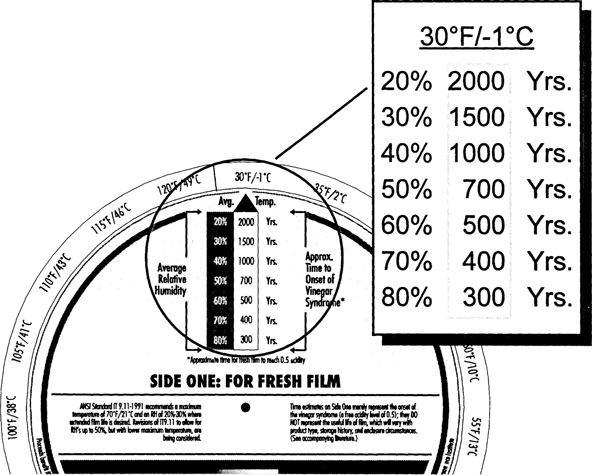
Fig. 7: Climate Index for Fresh Film at 30°F
The Wheel allows “trade-offs” to be determined to maintain specific CI values. Suppose that a CI of at least 600 years is desired. Fig. 6 shows that this CI value can be achieved at 40° F/30% RH. Suppose that you can't achieve 30% RH or that you want to keep the humidity very close to the conditions in the outer rooms in your building. A quick check of the Wheel shows that you can achieve a CI of 700 years at 50% RH if you keep the temperature at 30° F (see Fig. 5).
While Side One of the Wheel provides CI values for fresh film to reach 0.5 acidity (the brink of catastrophic deterioration), Side Two of the Wheel shows the CI values for film as it changes from 0.5 acidity to 1.0. To put acidity values into some sort of real-life perspective, it is handy to think of them in terms of the number of feet of film that contain one teaspoon of vinegar. Roughly 225 to 900 feet of fresh film having an acidity between 0.05 and 0.2 are required if we want to “extract” one teaspoon of vinegar. At 0.5 acidity, on the edge of the rapid autocatalytic stage, about 90 feet of film contains a teaspoon of vinegar. For 1.0 acidity film, the required length halves to about 45 feet. In the final stages of deterioration when film has an acidity of at least 5.0, nine feet or less of film will provide the equivalent of a teaspoon of vinegar. Even at this second 1.0 acidity endpoint, film is not at the end of its useful life.
Film starting with a higher acidity (0.5 versus “fresh film”) deteriorates at a faster rate. This faster deterioration rate is reflected in the CI values given on Side One and Side Two of the Wheel. At 70° F/50% RH partly degraded film has a CI of only five years compared with fresh film at 40 years (Fig. 8 and Fig. 5). In these early stages of deterioration, degrading acetate will benefit from improved storage conditions. Consider a drop in temperature from 70° to 40° F (Fig. 9). The CI value improves from 5 years to 50 years—a ten-fold increase. A further drop into cold storage at 30° F increases the CI even further to 110 years (Fig. 10). Values on this side of the Wheel represent somewhat maximum improvements for already degraded film. As the condition of the film base gets worse and worse beyond the 0.5 acidity level, thermal dependence of the degradation decreases and the autocatalytic nature increases.
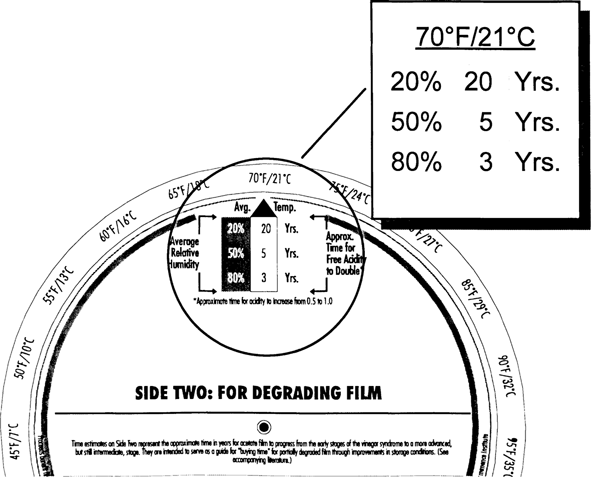
Fig. 8: Climate Index for Degrading Film at 70°F
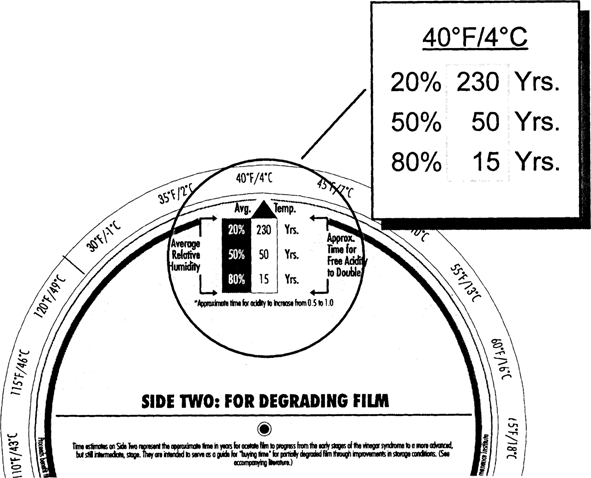
Fig. 9: Climate Index for Degrading Film at 40°F
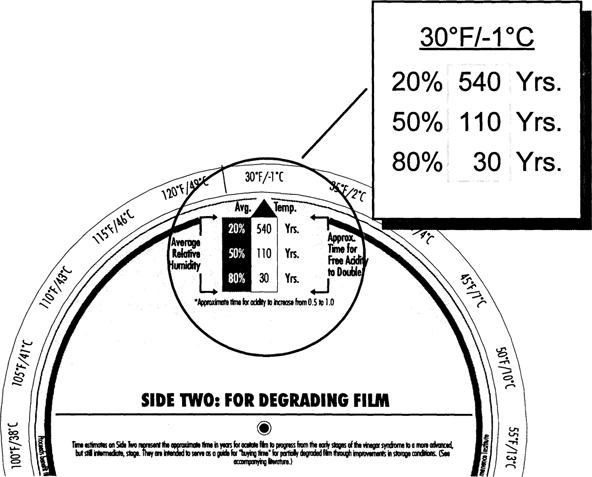
Fig. 10: Climate Index for Degrading Film at 30°F
One major advantage of the Guide is that the benefits of improved storage can at last be put in terms of dollars and cents. Since all materials will ultimately degrade, the film collection can be viewed as a declining investment. By having a quantitative measure of deterioration, the value of the collection can be amortized over the expected life of the film. A “cost per year” can be calculated under the current conditions and compared with a “cost per year” under improved conditions with the cost of improving the storage conditions factored in. Even from a very simplistic approach, benefits can be shown. Suppose that your collection has been valued at $50,000 and is currently stored at 70° F/50% RH. It costs $120,000 to upgrade your storage area to 40° F/50% RH. Your collection is losing value currently at a rate of $1250.00 per year. The cost of the improvement plus the value of the collection is $170,000, but the time period is much longer. Over the new CI of 350 years, the collection, including the cost of renovations, is losing value at a rate of $485.71 per year. These numbers are obviously not very realistic and don't include the cost of running the two vaults, but they do serve as an illustration.
Although the Guide is based on accelerated aging data, the general trends in data have been confirmed by experience. Evelyn Hofer, in New York City, for example, has totally lost a collection of her negatives from the early 1960's. The negatives were kept in a Manhattan apartment that was simply closed up and not occupied during the summer months. A number of other archeological and scientific institutions have had very similar experiences. If we assume that Hofer's negatives existed on average at roughly 80° F/50% RH, (hot and humid in summer and warm and very dry in the winter), the Wheel predicts a CI (to 0.5 acidity) of roughly 20 years. Thus 0.5 acidity would be expected to be reached in the early to mid 1980's. The next stage of degradation (from Side Two of Wheel) may take roughly another two years and final destruction (1.0 acidity to say, 10.0 acidity) may take another three years or so. This approximate accelerated data “predicts” that the film may reach ultimate destruction probably around 1990 ± 5 years. The film was first noticed in unprintable shape in 1992. This data is only approximate and should not be expected to closely predict collection life expectancies. It must be carefully noted that the Guide does not predict the life span of individual film samples but is a measure of the quality of the storage environment.
There are a number of implications of the predictions from the Guide:
The ultimate conclusion presented by the Guide is that an improved storage environment is the best overall answer to film preservation.
The author would like to thank the Division of Preservation and Access of the National Endowment for the Humanities, the National Historical Publications and Records Commission and Eastman Kodak Company for funding the research. Thanks also to Fuji Photo Film Ltd. and the Eastman Kodak Company for providing film samples used in the research as well as technical expertise. Finally, thanks to Mark McCormick-Goodhart for suggesting the application of his “Time Out of Storage” idea to our acetate data.