Topics in Photographic Preservation 2001, Volume 9, Article 8 (pp. 103-114)
The effects of conservation treatments on photographic material are a growing concern to many photographic conservators; this concern has been expressed in recent conference proceedings and journals*. Platinum photographs are particularly at risk to acid catalyzed degradation as a result of their fundamental process. The alternative process utilises ferric salts and acid clearing of the images. These two materials can be problematic and may cause the image-holding paper substrate to yellow, changing the visual relationships and impact of the image. Due to their physical composition, “traditional” paper conservation treatments have usually been considered appropriate. However, they have exhibited some unexplained reactions to washing. On occasion, when washed to reduce the discoloured degradation products, the pristine and inherent lustrous surface quality of the photograph has been irretrievably altered. This may very well be the result of a combination of effects: change in the surface quality of the paper, loss of materials important to the paper qualities (ie. sizing), and loss or changes in the platinum distribution. In this research project, I propose to study the effect of aqueous immersion washing times on the distribution of platinum metal in the substrate. This will be done with Energy Dispersive X-ray analysis of bulk samples with a Jeol® JSM 840 Scanning Electron Microscope equipped with a Quartz® PCI digital image acquisition and management system and a LINK® AN10000 x-ray and microanalysis instrument. The spectral data will be mathematically analysed and plotted with Microcal® Origin 4.1. The morphological and spectral information will be compared with densitometry interpretation gained by scanning samples with an UMAX® Powerlook II scanner with the image analysis program UTHSCSA® Image Tool and Image-Pro Plus version 4 from Media Cybernetics. Samples will be prepared according to the historical formulas used by photographers at the turn of the century, and will be artificially aged to replicate the characteristic yellow discoloration.
The platinum process was invented and patented by the Englishman Richard Willis in 1873, of the future Willis Platinotype Company of London. (Greenough et al. 1989). The process was further refined by Guiseppe Pizzighelli and Baron von Hvbl of Austria, with their results published in 1882 (Crawford 1979). Platinotypes are described as aesthetically encompassing image colour, depth, and tactile qualities to produce “a physical presence unmatched in photographic prints” (Gassan 1974). Platinum photographs offer a rich and uniquely long tonal range. Colour possibilities extended from cool blue-black to warm browns. The matte surface quality affords a decreased overall sharpness lending the images an atmospheric effect. Due to their unique and distinct aesthetic character platinum prints were especially popular with the pictorialist movement in photography existing at the turn of the century.
As explained by Greenough, Snyder, Travis and Westerbeck, these photographers promoted:
…‥an emotive photography that would appeal not to reason or the intellect, which were the properties of scientific enquiry, but to intuition….they favoured printing processes such as carbon, platinum, gumbichromate, and bromoil, as well as exotic combinations, which either in their initial application or in the development allowed for considerable reinterpretation of the negative. To intensify the expressive and tactile quality of their images they often hand-coated their own paper, which had been carefully selected for its size, weight and tooth. The pictorialists' manipulation of their prints was one of the most controversial aspects of the movement and it served many purposes. First, and most obviously, the size, tonal scale, print quality, and variety, even the texture, clearly separated pictorialists' work form that of the Sunday snapshooter. But the pictorialists' photographs also represented a rebellion against the growing standardisation of photography by large, commercial manufacturers. (Greenough et al. 1989)
Famous photographers, Alfred Stieglitz, Edward Steichen, Edward Weston, Peter Henry Emerson, Frederick Evans, Clarence H. White, F. Holland Day, Alvin Langdon Coburn, and Paul Strand all used platinum in their photographic oeuvre.
The aesthetics of these prints were not the only reigning factor in their overall popularity. Platinum proved to be a very stable image-forming metal (more so than silver). They were valued for their permanency because of their superior resistance to peroxides and other oxidising agents. The decline of this form of photography is attributed to evolving economics—the War Effort during World War I banned its use, which subsequently caused the price of the metal to skyrocket. While replacement metals were available such as palladium, strides in the commercial availability both financially and commercially of silver halide processes had a profound influence in platinum falling out of fashion. Currently, Platinotype photography is still practised and is used primarily as a fine art medium.
Platinotypes are an alternative photographic process based on the light sensitivity of iron salts. This ferric process involves the reduction of platinum salts to its elemental metal and the clearing of the residual iron salts with acid. The paper is sensitised with an aqueous solution of oxalic acid (H2C2O4), ferric oxalate (Fe2(C2O4)3·5H20) and sodium tetrachloroplatinite tetrahydrate (Na2PtCl4 · 4H2O) or potassium tetrachloroplatinite (K2Pt Cl4) (McCabe 1995). The chemical reaction is oxidation-reduction with the iron being reduced when exposed to ultraviolet light from ferric to ferrous salts (FeC2O4·2H2O) thus creating a light latent image. The image is then developed in potassium oxalate (K2C2O4·H2O), which oxidises the latent image to give the metal image. Residual iron deposits are cleared away with acid, in most cases hydrochloric acid is used (Gottlieb 1995).
The image forming metal is embedded in the paper structure and is considered a single-layer photograph (Hendricks 1991). When considering the permanency of platinotypes, they are often described as “exceptionally durable” due to platinum being considered “an element almost totally impervious to chemical attack…” (Time Life Books 1972). More accurately summarised, platinotypes are really only, “as permanent as the material on which it is printed” (Hafey and Shillea 1979). The deterioration of platinum prints is a result of the degradation of the paper support rather than the image metal. These photographs possess an amount of inherent vice as a result of their elemental process. Platinum metal is a catalyst for acidic decay and acceleration of paper embrittlement, as well as image transfer to adjacent sheets (Hess Norris 1985).
The iron basis of the process and the clearing of the prints in acid can prove disagreeable to the paper substrate. The processing in the acid clearing bath can be problematic in two ways. Improper clearing can leave iron salts embedded in the paper contributing to cellulose degradation by the oxidation of the iron, and residual acid contributes to acid hydrolysis chain-scission of the cellulose. Iron can encourage very harmful auto-oxidation of cellulose: if present in the form of iron (III) ions, electrons can be accepted from the cellulose fibres and thus oxidising the cellulose and forming damaging free radicals. The iron (III) ions are in the process reduced back to iron (II) ions, and in the presence of air, electrons are donated from the oxygen to oxidise iron (II) ions to iron (III)—creating cycle of cellulose breakdown (Gent and Rees 1994).
These factors, as well as improper handling, display and storage, place the platinum image in a vulnerable position—despite the nobility of the metal. Often the damage is observed as disfiguring yellowing or discoloration of the image bearing substrate. This discoloration can severely impact the aesthetic effect of the object and can even obscure the information within the image.
Because of the lack of emulsion layers conservation treatments are often directly borrowed from paper conservation. Surface cleaning, washing, repairs of mechanical damage to the paper substrate and other treatments common to paper conservation can be applied to platinotypes (Hess Norris 1985). Washing by aqueous means is a common strategy used to reduce discolouring degradation products in these objects. Aqueous immersion, float, and suction table techniques have been performed with distilled and/or deionized water. To simulate detergent like action, balanced alkali solutions such as sodium metaborate (Hess Norris 1980) have been used. More drastic washing treatments have involved the use of the chelating agent ethylene diamine tetra acetic acid (EDTA) in conjunction with sodium dithionite, (Gent and Rees 1995) and bleaching solutions such as calcium hypochlorite (Hess Norris 1980). Unfortunately it has been observed that washing treatments, even without chemical additives can unpredictably and irrevocably alter the surface quality of the photograph (Hess Norris 1995), resulting in a loss of the inherent lustrous character of the image.
Changes in the surface quality of these photographs as a result of treatment are recognised as a combination of effects including the loss or change in materials pertinent to the character of the paper sheet (i.e. sizing, fillers, etc.). This research project is concerned with the effects of aqueous immersion treatments on the image forming metal. These changes will be monitored and observed with scanning electron microscopy (SEM) and energy dispersive x-ray analysis (EDX), which will provide morphological and spectral information on the distribution of the image metal. This information will be subsequently compared with the densitometry interpretation of the samples by an image analysis program.
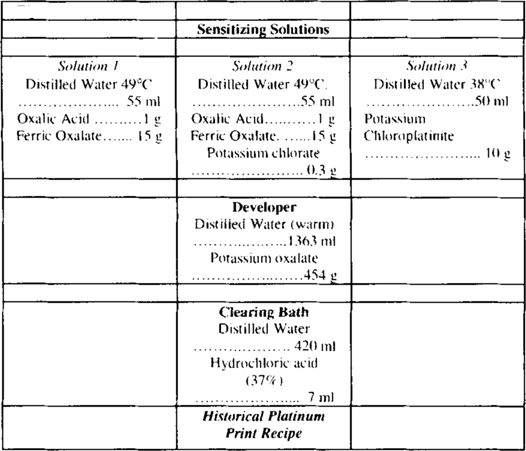
While using historical samples obviously allows for a more realistic approach, the decision to fabricate platinum samples was driven by two main concerns. The first being that the use of the data gained from the analytical techniques is comparative, and therefore required a simple, standard image design. The second concern was the possibility of finding historical platinum samples and being able to identify all the numerous possible chemical components. These concerns withstanding, platinum photograph samples were produced in accordance with historical recipes. Although there are many manipulations of the platinum process (Gottlieb 1995), it was decided to follow a simple recipe to reduce the number of variables to be considered (Ware 1998–1999). The recipe followed is found in William Crawford's 1979 publication The Keepers of Light: A History and Working Guide to Early Photographic Processes, and summarised in figure 1.

Figure 1
To further reduce the number of variables, solution 2, which acts to control contrast, was omitted. Solution 1 and 3 mixed to the ratio of 22: 24 drops was used to sensitise an approximately 8 × 10 inch area on a sheet of Cranes 901b wove natural white 100% cotton rag paper. The sensitizer was spread with a dampened squirrel bristle brush, and left to line dry for 20 minutes in a dark room. The sheet was exposed by contact printing for 27 minutes with a 250-watt Sylvania® heat lamp. The negative comprised of a 20.5 × 25.5 cm transparency of 11 gradual tonal steps from dark to white along the height. Once exposed the print is quickly immersed in the developer (a saturated solution of potassium oxalate) for 1 minute. The print is then cleared in three consecutive baths of hydrochloric acid for 5 minutes each. The print is finally washed in running distilled water for 20 minutes. The resulting photographic print is represented in figure 2.

Figure 2
One strip along the height of the image was removed before the photograph was artificially aged. Accelerated ageing of the samples was performed as per ISO 5360 standards. The object was conditioned to 65% relative humidity, placed in a heat resistant airtight enclosure and introduced to 80°C for 24 days in an Imperial III laboratory oven. Ageing temperature, duration and humidity levels were chosen to attempt to induce acid hydrolysis chain scission and iron oxidation in the paper substrate within the given time restraints (Ware and Nishimura 1998–1999). Successful degradation of the substrate occurred observed form the significant yellowing of the paper from a light natural cream to butter yellow. The degradation of the primary support was also superficially observed by recording the surface pH of the Crane's paper before sensitisation and after sensitisation and accelerated ageing was performed using a Fischer Acument® Model 230 pH/ION METER with an Orion® Ag/Ag Cl combination pH flat surface epoxy body non-fillable electrode. The surface pH of the paper dropped dramatically from 6.3 to 3.
Once aged, the sample was divided into eight equal strips along the tonal range to perform immersion aqueous washes of varying times from 30 minutes to 3 hours in 30-minute increments. The complete separation of the photographic sample is pictured in figure 3.

Figure 3
The washing of strips 3 through 8 was performed with distilled water obtained from a Polar Bear® Water Distillers Manufacture Co., (Alberta Canada) distillation tank. Each 3.0 × 19.0 cm strip was immersed as per standard paper conservation technique in their own water bath vessel to allow 1 litre of water to have action on the samples. Each strip was dried overnight face up on a blotter, and placed in protective archival PRINTFILE® negative sleeves.
A Jeol® JSM 840 Scanning Electron Microscope equipped with a Quartz® PCI digital image acquisition and management system and a LINK® AN10000 x-ray and micro analysis instrument were used to perform the SEM–EDX analysis. The compiled EDX spectra data was converted for mathematical use in Microcal® Origin 4.1 (plotting program with analysis tools, any use of these tools are denoted with an*).
EDX analysis was performed on tone 00, 02, 04, 06, 08, and 10 of the aged washed for 30-minute sample to verify the platinum response curve in the ascending tones. SEM and EDX Analysis were performed on tone 10 and tone 00 of each washed strip; aged and unwashed strip; and un-aged un-sensitised un-washed strip. EDX of the carbon coated 3mm circles of each sample was performed on an area represented by 50x magnification, with 1000 counts, a live time of 100 seconds, probe current of 2 × 10−9 amps (penetration approximately 3μm), accelerating voltage of 20 keV, and 8% dead time. Both Back scattered and secondary electron images of each tone 10 and tone 00 sample were captured at 500x magnification with the same conditions save the probe current at 2×10−10 amps. EDX and SEM analysis were also performed under the same conditions at a higher magnification (5500x, probe current 2×10−9 amps) to characterize the physical identity of the image forming metal as discrete particle, islands or a film.
Densitometry interpretation was performed with a UMAX® Powerlook II scanner at 600dpi optical with 8 bits 256 levels of grey, coupled with the image analysis program UTHSCSA® Image Tool. The sample strips were prepared for scanning by folding the bottom edge to reveal the aged washed Crane's paper and adding a strip of un-aged un-sensitised Crane's paper along the top edge as pictured in figure 4.
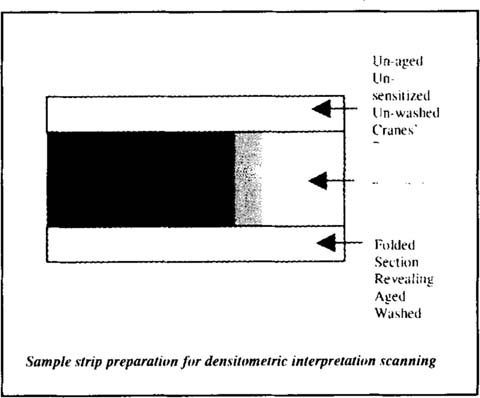
Figure 4
Platinum (atomic number 78) has a low platinum peak detectable between 2-3keV with a Mα principle emission line at energy 2.048 keV and less intense minor line Mβ at energy 2.127 keV with an intensity of (50). Platinum also has a higher peak detectable between 9–10 keV with a principle emission line Lα at energy 9.441 keV, and two lesser intense minor lines: Lβ1 at energy 11.069 keV with an intensity 50, and Lβ2 at energy 11.249 keV wit an intensity of (20). A sample platinum spectrum is pictured in figure 5.
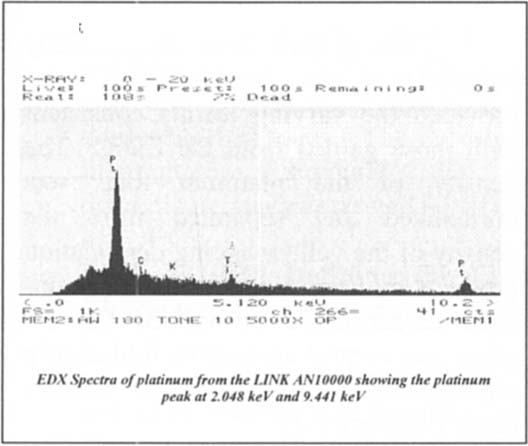
Figure 5
The EDX analysis performed on tones 2, 4, 6, 8, and 10 of the aged, washed for 30-minute sample to verify the platinum response curve in the ascending tones revealed the low platinum peak. Tone 00 of the sample revealed no platinum peak. This is demonstrated linearly and 3-dimensionally in figure 6 and figure 7.
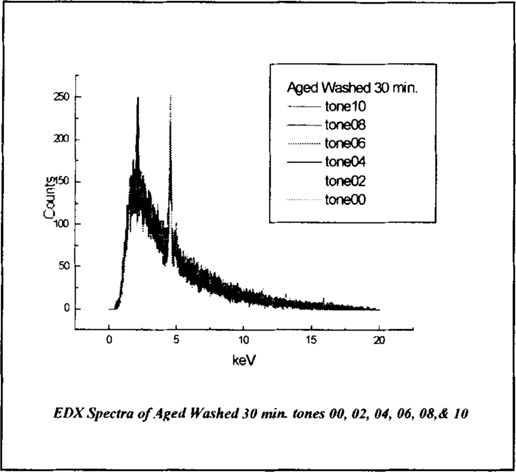
Figure 6
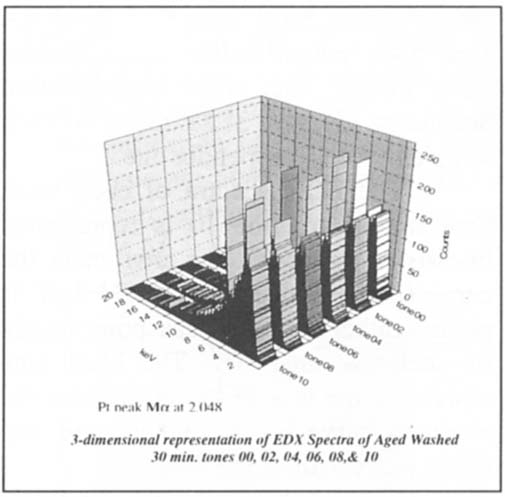
Figure 7
The response curve data is further represented by Gaussian fit* showing the increasing amount of platinum per each tone in figure 8.
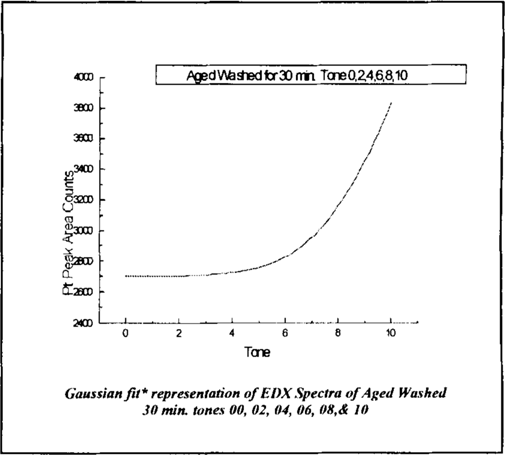
Figure 8
The complete EDX spectra, 50x magnification SEM image of the EDX area, 500x magnification Backscattered SEM image, and 500x magnification Secondary Electron SEM image of each sample is seen in appendix one.
The EDX spectra of each wash time sample of tone 10 is represented linearly in figure 9. The area under the curve (peak) between 1.94–2.34 keV is plotted rather than the peak point height for inclusive analysis. The black line represents the plot of the area under the platinum curve (peak) and the red line offers a linear fit of the data.
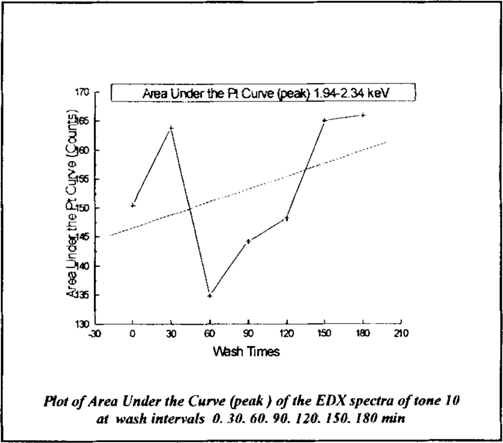
Figure 9
The peak heights semi-quantitatively represent the amount of platinum present. Error incurred by inconsistent development of the photograph cannot be calculated but must be factored. The changes in the amount of platinum between wash times between 0 and 60 minutes surpass that which could be attributed to uneven development. The inconsistencies perhaps can be explained by the intense and possibly irregular action of the immersion treatment removing and re-depositing platinum in the first hour of treatment. Doing the same analysis to tone 00, pictured in figure 10, it was revealed that changes in platinum distribution are marginal, but there is an increase in platinum in the tone where there should not be any.
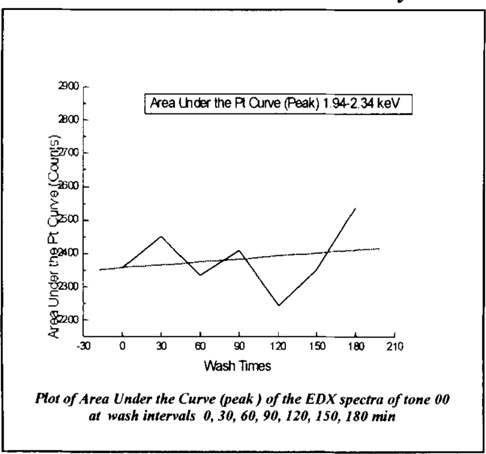
Figure 10
Data gained from densitometry interpretation of the samples also provided the varying results consistent with those gained from the EDX. The density of the platinum tone was normalized and separated from the density of the yellow ageing degradation products by using the density of the un-aged un-sensitized un-washed Crane's paper as a control against the folded strip representing the aged washed Crane's paper (see figure 4).
The difference between the two was added to the density of the tone (as the greater the number the whiter the tone). figure 11 shows how the densitometry interpretation of tone 10 for strips Aged Un-washed through Aged Washed 180 min. coincides with the area under the platinum EDX curve (peak) plotted in figure 9.
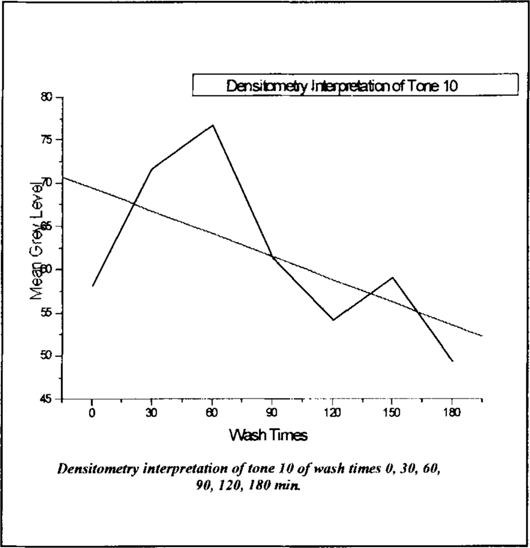
Figure 11
The plot of the density reveals the same considerable variances in the platinum presence/distribution in the first hour of treatment seen previously in the EDX data. The higher the Mean Grey Level number is on the densitometry interpretation graph, the lower the point on the Area Under the Curve graph should be. High grey level numbers indicate lighter/whiter tones, and low grey level numbers indicate denser/blacker tones. The densitometry interpretation supports the semi-quantitative analysis gained by EDX.
Further exploration with SEM and EDX analysis was prompted by the varying results above. Both the Back Scattered and Secondary Electron SEM images did not reveal platinum in the anticipated islands of metal between the interstices of the fibres. While platinum particles singularly are too small to detect with EDX (Barger 1998–1999), conglomerates of the metal should be detectable, as the metal in platinum photographs is repeated referred to as existing embedded in the interstices of the fibre in the literature. Morphological observations found that the fibre interstices appear clear of any large or discrete deposits of material. figure 12 typifies the character of the photograph in tone 10.
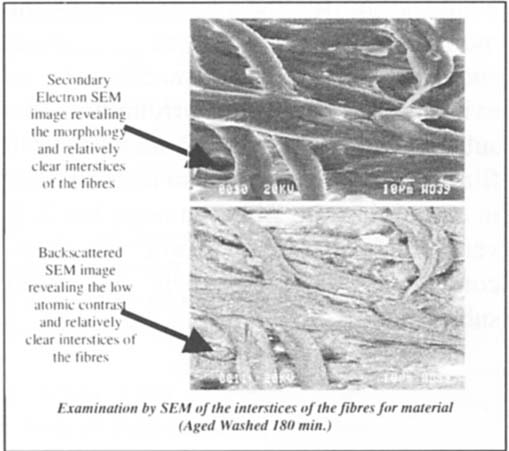
Figure 12
EDX and SEM analysis was performed on and off particles (heavy and light elements respectively) detected with Backscattered SEM at the higher magnification of 5500X. The points of analysis are marked in figure 13. Both of the resulting EDX spectra are seen in figure 14.
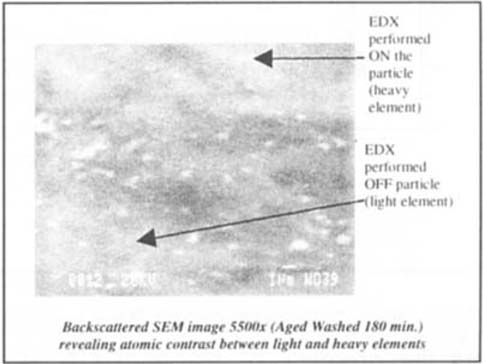
Figure 13
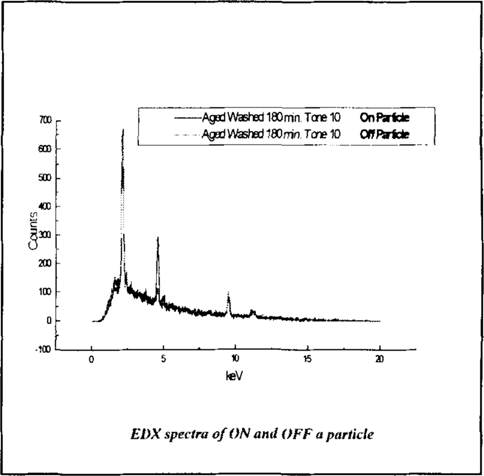
Figure 14
The EDX spectra show a marked amount of platinum is present ON the particle and OFF the particle. This lead to the examination of the amount of platinum present OFF the particle on randomly selected tone 10 samples compared to a tone 00 of a sample (a sample with no platinum). The resulting EDX of OFF particle locations of the Aged Un-washed, Aged Washed 180 min., Un-aged Un-washed tone 10 samples is pictured in figure 15.
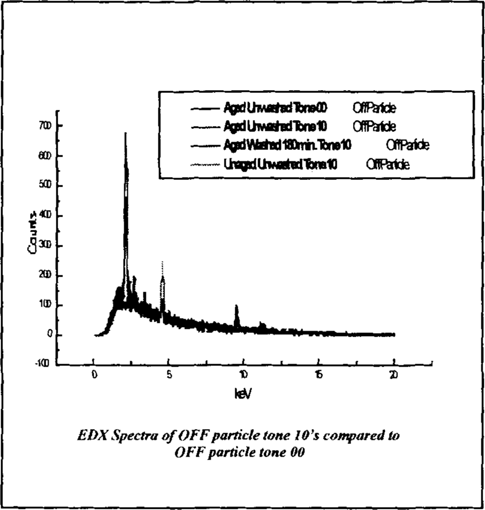
Figure 15
The morphological observations and the presence of platinum ON and OFF the particles found at 5500x magnification indicates that platinum does not exist between the interstices of the fibres but as a coating of the fibres. In essence a film rather than discrete islands of materials as previously understood.
Considering the relationship of the platinum distribution to the wash times, it is revealed that the platinum is affected by the immersion wash. While the effects are not conclusively extractable as a loss, redistribution of the platinum within the paper substrate appears to be occurring. It can be concluded from the results that the disturbance of the platinum occurs most dramatically and erratically (both a loss and increase of platinum unpredictably) within the first hour of immersion washing. This unpredictable nature could be attributed to the existence of the platinum metal as a film on the fibres rather than as previously understood discrete islands between the interstices of the fibres. By existing as a film on the fibres the metal is vulnerable to the reactive expansion and shrinkage of the paper fibres. The fibres react to the immersion treatment and do not necessarily regain their original pre-treatment state. The disturbance to the fibre results in both losses of platinum: by fissures, cracks, breaks and thinning of the film as the fibre swells and distorts; and as increases in platinum: from the overlapping, re-embedding and condensing of the film as the fibre shrinks, and contracts. The characterization of the image forming metal as a film and not discrete particles increases the understanding of the unpredictable nature of platinum to immersion washing.
Further research into the changes occurring to the platinum metal as a result of immersion treatment would benefit from the use of transmission electron microscopy (TEM). TEM's capability for higher magnifications would allow for the observation of the morphology of the platinum particles and possibly their mechanics in existence as a film surrounding the substrate fibres. AUGER analysis' thin film capabilities would also be beneficial in examining the film character but it is very difficult to perform on non-conductive samples such as paper substrates.
Barger, Susan. 1998–1999. Personal communications. Santa Fe, New Mexico
Bunnell, Peter C. ed. 1984. Nonsilver printing processes: Four selections, 1886–1927. New Hampshire: Ayer Company, Publishers, Inc.
Bostick, Melody 1998–1999. Personal communications. Santa Fe, New Mexico
Coe, Brian and Mark Haworth-Booth 1983. A guide to early photographic processes. London: Hurtwood Press in association with the Victoria and Albert Museum.
Crawford, William 1979. The keepers of light: A history and working guide to early photographic processes. New York: Morgan and Morgan.
Davidson Lowe, Sue. 1983. Stieglitz: A memoir/biography. New York: Farrar Straus Giroux.
RIT 1972. First all-union conference on non-silver and unusual photographic processes, Kiev. New York: Graphic Arts Research Centre Rochester Institute of Technology.
Fraser, Jeff 1998–1999. Personal communication. National Research Council of Canada, Ottawa, Ontario.
Gassan, Arnold 1974. A handbook for contemporary photography, 3rd ed. Ohio: Handbook Company.
Gent, Megan and Jacqueline Rees 1994. A conservation treatment to remove residual iron from platinum prints. The Paper Conservator 18: 90–95
Gottlieb, Adam. 1995. Chemistry and conservation of platinum and palladium photographs. Journal of the American Institute for Conservation. 34 (1): 11–32.
Greenough, Sarah, Joel Snyder, David Travis, and Colin Westerbeck 1989. On the art of fixing a shadow. (Exhibition at the National Gallery of Art and The Art Institute of Chicago). Boston, Toronto, London: Bullfinch Press/Little Brown And Company.
Hess Norris, Debbie 1980. Eakins at Avondale: Discovery, examination, and treatment of his platinum prints”. The American institute for conservation of historic and artistic works preprints of papers presented at the eighth annual meeting, San Francisco, California, 22–25 May.
Hess Norris, Debbie 1985. Platinum photographs: Deterioration and preservation. Photographi Conservation. 7 (2): 1.
Hess Norris, Debbie 1992. Photographs. Caring for your collections. New York: Harry N. Abrams, Inc., Publishers.
Hess Norris, Debbie 1995. Current research needs in the conservation treatment of deteriorated photographic print materials. Research techniques in photographic conservation, Copenhagen 14–19 May. Hess Norris, Debbie. 1998–1999. Personal communications. University of Delaware.
Hendricks, Klaus. B 1991. Fundamentals of photographic conservation: A study guide. Toronto: Lugus.
Howell-Koehler, Nancy 1980. Photo art processes. Massachusetts: Davis Publications, Inc.
Kosar, Jaromir 1965. Light-sensitive systems: Chemistry and application of nonsilver halide photographic processes. New York, London, Sydney: John Wiley and Sons, Inc.
McCabe, Constance 1995. Understanding Alfred Stieglitz' platinum and palladium prints: Examination by x-ray fluorescence spectrometry. Research techniques in photographic conservation, Copenhagen 14–19 May.
Nadeau, Luis 1989. Encyclopaedia of printing, photographic and photomechanical processes. Vol. 1 and 2. New Brunswick: Atelier Luis Nadeau.
Nadeau, Luis 1997. History and practice of platinum printing, 3rd rev. ed. Fredericton, New Brunswick: Atelier Luis Nadeau.
Neblette, C.B. 1962. Photography: Its materials and processes. New York: Van Nostrand Reinhold Company.
Newhall, Nancy 1975. Edward Weston: Flame of recognition, his photographs accompanied by excerpts from the daybooks and letters. New York: Aperture, Inc.
Nishimura, Douglas 1998–1999. Personal communications. Rochester, New York.
Rempel, Siegfried 1987. The care of photographs. New York: Nick Lyons Books.
Severson, Douglas. G. 1995. Alfred Stieglitz' palladium photographs and their treatment by Edward Steichen“. Journal of the American Institute for Conservation. 34 (1): 1–10.
Shillea, Thomas John 1984. The platinotype- A brief history. PhotographiConservation Vol. 6 (4): 1, 4.
Shillea, Thomas John and John Hafey 1979. The platinum print. Rochester, New York: Graphic Arts research Centre Institute of Technology.
Sullivan, Richard 1998–1999. Personal communications. Santa Fe, New Mexico.
Thurgood, Brian Personal correspondence. National Archives of Canada, Gatineau, Quebec.
The Editors of Time Life Books. 1972. Life library of photography caring for photographs: Display, storage, restoration. New York: Time life Books.
Tani, Tadaaki 1995. Photographic sensitivity: Theory and mechanisms. New York and Oxford: Oxford University Press.
Ware, Michael 1998–1999. Personal communications. Derbyshire, UK.
Willsberger, John 1977. The History of photography. New York: Doubleday and Company, Inc.
1992. The imperfect image: Photographs, their past, present and future. Centre for Photographic Conservation International Conference at Low Wood Conference Centre, Windermere.