Topics in Photographic Preservation 2009, Volume 13, Article 28 (pp. 209-220)
Presented at the 2009 PMG Winter Meeting in Tucson, Arizona
Establishing protocol for surface cleaning inkjet prints is a need within the field of art conservation, and was determined to be the goal of this project. The materials tested are widely used to create fine-art inkjet prints. The Epson Stylus Pro 4800 printer was chosen for this reason, and because it employs the K3 Ultrachrome ink set, which is pigment based. Two different microporous inkjet papers, Epson Ultra Premium Photo Paper Luster and Ilford Galerie Gold Fibre Silk , were tested. A digital file, or “target,” was designed using well-known color space values, and used to create twelve sample prints. Preliminary quantitative measurements of changes in color were made, as well as qualitative analysis of surface sheen and dye migration. This analysis was repeated after testing.
The cleaning solutions tested were deionized water, a water and ethanol solution, and a PhotoFlo and water solution. These materials were chosen because they are common cleaning agents which are likely to be found in the labs of most photograph conservators. Two cleaning techniques were tested; application with a cotton swab and full immersion.
Results suggest that two of the cleaning materials used may be acceptable for future treatment of inkjet prints. Ethanol and water was determined to be inappropriate for cleaning the inkjet materials tested in this study, and immersion was determined to be inappropriate for prints created on Ilford Galerie Gold Fibre Silk. Cleaning with cotton swabs produced noticeable changes in surface sheen in paper white areas, but this technique may be acceptable for image areas. Further research of alternative cleaning methods and materials is suggested to yield conclusive results.
The research presented in this paper was conducted as an honors senior thesis project at the University of Delaware over the course of the 2008-2009 academic year. The thesis work began with a summer internship at Aardenburg Imaging and Archives that provided a foundation in creating and identifying inkjet prints, as well as an understanding of color science and color management. Over the course of the fall semester materials to test were determined, the target image was designed, the samples were created, and preliminary analysis was conducted.
After investigating the many different dry and wet cleaning methods currently employed by conservators when caring for photographs and works of art on paper, it was determined that this project would focus on wet cleaning. This type of cleaning was appropriate for the project because it provided the opportunity to test potentially damaging techniques on samples, rather than real art objects.
When selecting the materials to be tested it was determined to choose materials which were, and are, widely used to create fine-art inkjet prints. An Epson Stylus Pro 4800 printer was chosen for this reason, and because it employs the K3 Ultrachrome ink set.
The K3 Ultrachrome ink set is pigment based and consists of eight different color cartridges, including cyan, magenta, yellow, light versions of cyan and magenta, two greys and black. Two options for the black cartridge are available; either matte black or photo black. The black ink cartridge can be interchanged depending on whether matte or glossy paper is being used. For this project, the photo black ink was used.
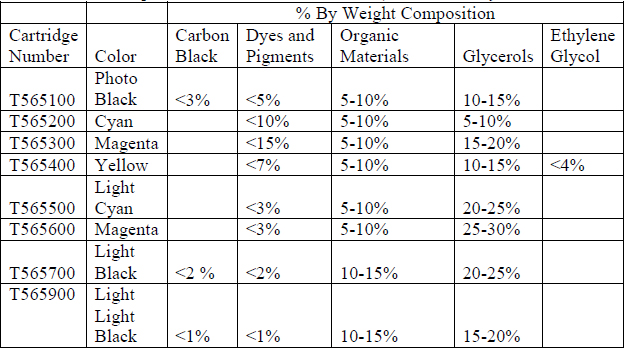
While the exact chemical composition of the K3 Ultrachrome ink is proprietary, its general composition is available (Table 1) (Material Safety Data Sheets 2000-2009). A pigment based ink set was chosen, rather than a dye based ink set, for several reasons. First, pigment based ink does not exhibit short-term drift like dye based ink. Therefore, using the K3 Ultrachrome ink eliminated the issue of deciding when the ink had finished “settling.” This was particularly important to this research in ensuring that any color change or dye migration would be due to the tests performed not independent movement of the ink after printing or fluctuations in humidity. Secondly, pigment based inks have proved to be more waterfast than dye based inks because pigment particles are not soluble in water. This ink set in particular has high waterfastness and permanence ratings according to the findings of Wilhelm Imaging and Research (Epson Stylus Pro 4800-Print Permanence Ratings (preliminary) 2005).
Table 1 Composition of K3 Ultrachrome Ink (Material Safety Data Sheets 2000-2009)

The final material chosen was paper. It was decided that two different microporous inkjet papers would be tested to compare whether or not the ink behaved differently on different paper supports. Microporous papers are a major type of paper being used by printmakers today, and were expected to respond better to wet cleaning in comparison to other inkjet papers. Two papers from the same general category were chosen so that they could be compared more easily, and provide more conclusive results. The two papers chosen were Epson Ultra Premium Photo Paper Luster and Ilford Galerie Gold Fibre Silk. While both are microporous papers, the Epson is a resin-coated paper, and the Ilford is fiber based. Because of this, the two papers were expected to respond to treatment differently. Both have “luster” type surfaces.
Epson Premium Photo Paper Luster was recently renamed by Epson, and is now called Epson Ultra Premium Photo Paper Luster. This paper has received high permanence ratings when used in combination with the Epson Stylus Pro 4800 and K3 Ultrachrome ink set according to tests conducted by Wilhelm Imaging and Research (Epson Stylus Pro 4800-Print Permanence Ratings (preliminary) 2005). This paper was also recommended by Mark McCormick-Goodhart as one likely to be encountered by conservators in the future due to its popularity among fine-art printmakers.
Ilford Galerie Gold Fibre Silk, in comparison to the Epson paper, is a fairly new fine-art inkjet paper. Little permanence data is currently available on this paper, and there are no waterfastness tests yet available. Although it is a fiber based paper, the back of the paper is coated with an anti-curl layer. Ilford Galerie Gold Fibre Silk also differs from the Epson paper in that it contains a baryta layer. This paper was chosen according to the advice of Mark McCormick-Goodhart and Martin Jürgens. Both McCormick-Goodhart and Jürgens expect inkjet papers containing a baryta layer to become popular among printmakers in the near future because of the way in which the baryta layer allows the inkjet paper to imitate the look of traditional photographic papers.
Figure 1 Target Design
Once the materials to be test were identified the next step was to design the target (Fig. 1). A small image was included to give a rough idea of how a real print would respond to cleaning. The hope was that this would provide the opportunity to make conclusions about the effects on real-world images. This information could then be used to guide future research decisions.
One of the first criteria for designing the target was use of a Minolta CR-400 chromameter. The color patches needed to be large enough to accommodate the instrument available, which meant they needed to be approximately one inch square. One inch of space was left between each color patch, so that if any dye migration occurred during wet cleaning, the colors would not interfere with one another or at least this effect would be minimized as much as possible. The amount of color patches was chosen in order to make the best use of space on a letter sized piece of paper.
After consulting with experts in the field including Mark McCormick-Goodhart and Martin Jürgens, it was determined to base the color patches off of the Greytag Macbeth Colorchecker colors. The exact L*a*b* numbers for these colors that appear on the Colorchecker are available on the internet (ColorChecker Colormetric Data 2008). The first row of the target contains the cyan, magenta, and yellow values of the Greytag Magbeth Colorchecker. The values of the black were set to the maximum black, or zero for every channel to reflect the darkest black the printer could create and to exclude the presence of other color inks as much as possible. The second row consists of red, green, and blue color patches. These colors were chosen to represent mixtures of the ink colors other than black. The third row consists of “dark skin,” “light skin,” and “neutral 6.5 (or grey)” color patches to represent how skin tones and neutrals would react. The fourth row color swatches consist of a checkerboard pattern to make qualitative analysis of potential dye migration easier to perceive. This type of test swatch pattern has been successfully used by Mark McCormick-Goodhart for testing waterfastness of inkjet materials (McCormick-Goodhart and Wilhelm 2003).
Figure 2 Checkerboard Pattern
Each target was labeled along the bottom edge with the printer name, ink set name, paper type, date, and sample number so that the samples could be properly identified during all stages of the project (Fig. 1).
After the target design was complete, the sample prints were created. Photoshop CS3 was used in conjunction with the Epson driver. Image size was set to 8 bits/channel. In the Photoshop driver, color handling was set to “Photoshop manages color,” and appropriate ICC profiles were applied to the samples. The Epson Ultra Premium Photo Paper Luster profile was available on the computer used to print, while the Ilford Galerie Gold Fibre Silk profile had to be downloaded from the Ilford website. The rendering intent was relative colorimetric. In the Epson driver, the paper setting used for the Epson paper was Premium Luster Photo Paper, while for the Ilford it was set to Premium Semigloss Photo Paper as recommended by the manufacturer (Ilford Galerie Gold Fibre Silk 2009). The quality was set to 1440 dpi, or dots per inch, to obtain a high quality image. High speed was turned off and color management was turned off, based on common practice. Twelve samples were created and tested.
After the samples had been printed successfully, preliminary colorimetric measurements were made for each sample. This was done using a Minolta CR-400 Chromameter. Three consecutive measurements of each color patch were taken on each sample print. The paper white of each sample print was also measured. This was done in the same place, between the lower left corner of the thumbnail image and the upper right corner of the yellow checkerboard-patterned patch.
It was determined that analysis of change in surface sheen would be qualitative rather than quantitative due to the equipment available and the large amount of figures generated by the colorimetry measurements. In order to accomplish this, the samples were placed on a copy stand, and the lights were arranged at the most shallow angle allowed by the stand, which was 35o. The camera was then placed opposite the light source at approximately the same angle. This procedure was determined by consulting Harold and Hunter’s The Measurement of Appearance (1987) and several publications by Jonathan Arney, a leading researcher in the field of surface analysis (2002, 2006, 2007, 2007). Dye migration was also documented qualitatively only in the horizontal direction.
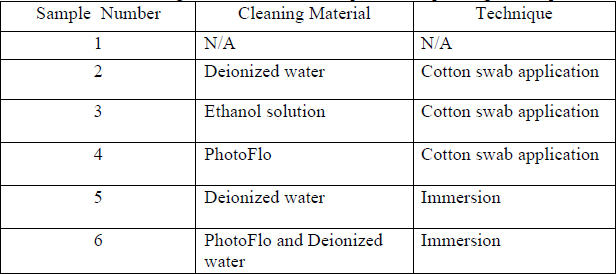
The three cleaning solutions tested in this research were deionized water, a water and ethanol solution, and a PhotoFlo and water solution. These materials were chosen because they are common cleaning agents which are likely to be found in the laboratories of most photograph conservators.
For this project, first a 70% ethanol 30% water solution was used, but this immediately proved to be extremely detrimental to the prints. Because of this, the 70% ethanol solution was only tested on the cyan patch of each sample, and the rest of the testing was conducted using a 30% ethanol and 70% water solution.
The particular brand of PhotoFlo used was Kodak Professional Photo-Flo 200 Solution. For this surface cleaning test, PhotoFlo was mixed with water according to the manufacturer recommended ratio, which is 1 part PhotoFlo to 200 parts water.
Two cleaning techniques were tested; application with a cotton swab and full immersion. Manufactured swabs are of uniform shape and size in comparison to handmade swabs, and therefore were appropriate for this research project.
Full immersion was tested during this project to provide a basis of comparison for the swab testing, and how the prints would react if exposed to maximum amounts of the cleaning materials. If the prints could withstand this test, exposure to lesser amounts of the cleaning materials would be likely to be successful. Studies on the Washing of Paper: Part 2: A Comparison of different washing techniques used on an artificially discoloured, sized paper by V. Daniels and J. Kosek was consulted when developing the treatment criteria for the immersion tests (2004).
The surface cleaning protocol developed was deliberately methodical and detailed in order to be easily reproducible and potentially used in future research. Pip Morrison’sSolvent Effects on Silver Dye Bleach Materials (2005) and Paul Messier and Timothy Vitale’s Effects of Aqueous Treatment on Albumen Photographs (1994) were consulted when determining treatment protocol. Table 2 below provides a general overview of the materials and techniques tested on each specific sample. Please note the same tests were conducted on the two different papers on samples with identical numbers. For example, both Ilford 1 and Epson 1 were used as controls.
Table 2 Cleaning Materials and Techniques Corresponding to Samples
During the swab application tests, the samples were placed on a blotter work surface. Commercially available non-sterile 6” cotton tipped swabs were used. Approximately 12 mL of the cleaning solution was placed in a 50 mL beaker. A cotton swab was dipped into the cleaning material perpendicular to and touching the bottom of the beaker. The amount of liquid in the beaker allowed the entire cotton swab to be immersed. The swab was then removed and rolled over a small blotter to remove any excess liquid. The dampened cotton swab was then immediately used to clean the surface of the sample print. On each sample, each color patch was cleaned with one swab, and three swabs were used to clean the small image. Therefore, a total of 18 swabs were used on each sample. The cleaning technique for each patch was kept as uniform as possible. Within each color patch, an area was cleaned using three counterclockwise circular motions of the swab, and this technique was repeated in an adjacent area within the patch until the entire color patch had been cleaned. The same speed was maintained and as little pressure as possible was applied when cleaning each patch. Six samples were treated in this way.
Immersion tests were conducted on four samples. It was determined that due to the dramatic results achieved using the ethanol and deionized water solution during the swab application tests, that the immersion test would not be conducted using an ethanol and deionized water solution. To conduct the immersion tests, the same procedure was used for both deionized water and the PhotoFlo and deionized water solution. Approximately 2 liters of cleaning solution were poured into an enamel tray. The pH was measured using pH test strips, and found to be neutral, or around 7, for both cleaning solutions. A sheet of Holitex, or spun-polyester webbing, was placed in the bottom of the tray as a support for the samples when removing them from the trays. The samples were then placed in the trays by bellying them onto the water surface and lowering opposite corners slowly. Each sample was completely immersed in the solution by rolling a glass stirring rod over the surface of the sample in the horizontal direction. The total immersion time for each sample was 15 minutes, and the solution was agitated approximately every 5 minutes. After 15 minutes had passed, the samples were carefully removed and allowed to air dry on a plastic screen.
During the cleaning tests, several observations were made which suggested that two of the surface cleaning materials and methods used were acceptable for future treatment of inkjet prints. Colorimetric, surface sheen, and dye migration assessment were conducted to aid in the evaluation of the materials and methods tested. Overall the results suggest that an ethanol and water solution is not appropriate for cleaning inkjet prints made with K3 Ultrachrome ink, and that deionized water alone or mixed with PhotoFlo may be potentially useful. Further research is suggested in various areas, including surface cleaning tests with other inkjet materials, other cleaning materials, and other cleaning methods.
While conducting cleaning with cotton swabs, it became immediately apparent that the ethanol and water solution is not an appropriate cleaning material for the inkjet materials tested in this study. An unacceptable amount of pigment was removed from the print surface by the ethanol cleaning solution when applied with cotton swabs (Fig 4.1). The action of the swab also served to displace pigment on the surface, giving the color patches an uneven, cloudy appearance. These effects were more pronounced in the Ilford sample (Fig 4.1). The samples which were cleaned with a swab using deionized water and PhotoFlo exhibited a small amount of pigment removal in comparison to the prints cleaned with ethanol and water. During immersion testing, no loss of color could be visually observed, which was promising. The Epson samples, however, exhibited misplaced pigment particles on the surface of the print, which suggested that some pigment had indeed been removed during cleaning, and was dispersed in the bath to some degree.
Figure 3 Cotton Swabs Used to Clean Ilford 3 with Ethanol and Water
Once the samples had dried thoroughly, after treatment colorimetric measurements were made. This was done in the same manner as the before treatment measurements; three consecutive measurements of each color patch were made and averaged. The ΔE value for each color patch was then calculated. ΔE is defined as the color change between two samples, using the CIEL*a*b* color space. The formula used to calculate this value is as follows:
ΔE= [(ΔL*)2 + (Δa*)2 + (Δb*)2] ½
When comparing ΔE values to observable color change, a ΔE of 1 signifies a visual color change. However, this value does not account for which color channel in particular has been altered, so the separate changes in L*, a*, and b* values must also be considered (Berns 2000).
Table 3 Calculated ΔE Values for Epson Samples
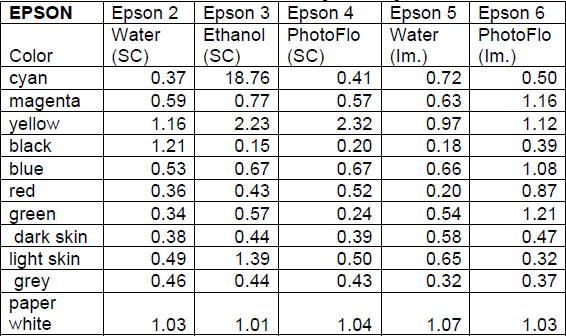
Table 4 Calculated ΔE Values for Ilford Samples
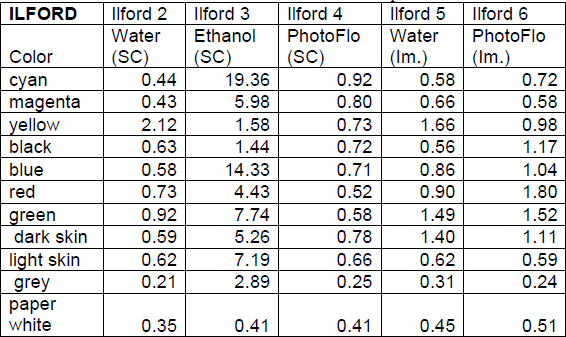
Swab cleaning tests conducted using deionized water and the water and PhotoFlo solution generally resulted in a ΔE of less than one observed for most of the patches (Table 3 and 4). Notable exceptions to this among the Epson samples include the black, paper white, and yellow patches after the deionized water cleaning test and the paper white and yellow patches after the PhotoFlo cleaning test. Among the Ilford samples, the green and yellow patches had a ΔE greater than one after the water cleaning test and the same can be said for the cyan patches after treatment with PhotoFlo. The ΔE for the Ilford sample cleaned with ethanol were much more dramatic and represent unacceptable change for conservation treatment; only the paper white patch had a value of less than one, and eight patches produced ΔE’s of greater than one. On the Epson sample, the light skin, paper white, and yellow patches had ΔE’s of slightly more than one. Though colorimetry results are less dramatic than those observed on the Ilford sample, visual analysis of the color patches shows that ethanol is still an unacceptable cleaning material for the Epson paper. The removal of color was distributed unevenly within the color patches.
Visual analysis suggested a lack of color change during the immersion testing, yet the after treatment colorimetry results revealed that there were visually noticeable color changes. The Epson sample immersed in water showed a ΔE of about one for the paper white and yellow patches, while the sample immersed in the PhotoFlo and water solution showed a ΔE of one or greater for the blue, green, magenta, paper white, and yellow patches. On the Ilford samples, the sample immersed in water exhibited this change in the dark skin, green, and yellow patches, and the sample immersed in PhotoFlo showed a ΔE greater than one for the black, blue, dark skin, green, red, and yellow patches. These changes were perhaps more difficult to detect because the removal was gradual and the pigment was dispersed in the solution. The immersion test results in the area of color change did not fail dramatically, which indicates that these solutions may be used successfully if different cleaning techniques are tested.
After comparing the calculated ΔE values among the various tests, consistent changes in yellow could be noticed in almost every sample. This was unexpected because the yellow patches did not appear to be changing more than the other test colors during the cleaning process. This suggests that minimal removal of yellow pigment results in a greater color change for yellow than in comparison to other colors tested. The one exception to this trend is the immersion test with PhotoFlo conducted with the Ilford paper. The change in yellow is much lower than with any of the other samples. Although this suggests that PhotoFlo immersion would be successful with the Ilford paper in terms of color change, this test proved unsuccessful in terms of change in surface sheen.
Another unexpected result was the lack of removal of ink from the black color patches. The only cleaning test that removed black ink was deionized water applied with a cotton swab on the Epson paper. Since the black colorant used is carbon black, generally considered to be soluble in water, it was expected that more ink removal would occur. The paper white colorimetry measurements of the Epson samples after treatment were also unexpected, especially since the Epson paper exhibited less of a change in surface sheen than the Ilford paper overall.
The surface sheen was altered by swab cleaning in a majority of the samples, especially in the paper white areas. The paper white areas became considerably glossier after treatment. This is particularly noticeable in the paper white area which was spot cleaned on each sample. Overall, the surfaces of the Ilford samples became noticeably glossier regardless of the cleaning material used. This is probably due to the differences in composition between the two papers, most likely the presence of a baryta layer. The Epson paper showed less of an alteration when cleaned with the deionized water and the PhotoFlo and water.
The results of the immersion tests in regard to surface sheen varied greatly between the two papers. The Ilford paper exhibited no change while immersed, but as the paper dried, it curled and fine cracks developed across the entire surface in a horizontal pattern. The surface also appears to have become somewhat glossier, but this is difficult to determine because of the cracks. This occurred after immersion in deionized water and the water and PhotoFlo bath. This dramatic change in surface sheen may also be due in part to the presence of the baryta layer. The Ilford paper was expected to dry differently from the Epson paper, because it is fiber based. Since the fibers absorb water and swell, and then contract during drying, this causes the paper to curl. This behavior is not exhibited by resin-coated papers, because the polyethylene layer prevents water from being absorbed by the fibers.
The Epson paper exhibited tidelines in some paper white areas prior to drying and the paper white areas became glossier after treatment. Visual comparison to the control sample and the calculated ΔE value of the paper white area support this conclusion. The paper white areas for both immersed samples show ΔE values of about one. This is consistent with the ΔE of the paper white areas of the swab cleaned samples, which certainly show a change in surface sheen.
In each of the samples, there was no visible evidence of dye migration occurring across the surface of the paper, regardless of cleaning solution and technique. This is not surprising, because the inks used were pigment based, and therefore the colorant particles would not be easily mixed with water and moved across the surface of the paper. While some of the samples which were swab cleaned exhibit movement of pigment on the checkerboard patterned color patches, this was caused by the movement of the cotton swab rather than action of the solvent on the pigment. The dye migration results are promising, as they suggest that wet cleaning may not be entirely destructive, and when applied to these materials will not cause the ink to bleed. This study did not assess whether or not downward migration (versus horizontal migration) occurred into the paper substrate. Such analysis could be conducted by microscopic examination of cross sections of the samples and this type of analysis is suggested for future research.
Changes in color, surface sheen, and dye migration serve as indications of whether or not an inkjet print has been altered by cleaning tests. Some techniques and materials were initially recognized as inappropriate due to the drastic alterations incurred, while others may require further research to determine whether or not the materials and methods used can be applied to surface clean actual art objects made from inkjet materials.
The use of ethanol solutions in wet cleaning inkjet prints created with K3 Ultrachrome Ink print on Epson Ultra Premium Photo Paper Luster and Ilford Galerie Gold Fibre Silk paper is unadvisable; removal of the ink from the print surface was instantaneous and severe. Despite the change in concentration of ethanol, and despite the less dramatic colorimetry readings for some of the patches, the pigment continued to be removed in a manner that made the color patches uneven in color. For this reason alone, it can be said with certainty that an ethanol and water solution is an unacceptable for cleaning for inkjet prints created with the materials tested in this study.
While cleaning with cotton swabs produced noticeable changes in surface sheen in paper white areas, this technique may be acceptable for image areas. Although some pigment was removed during testing with the deionized water and PhotoFlo and water solutions applied with cotton swabs, the overall affect on the color was barely visible. This is supported by the fairly low ΔE values, with the exception of the yellow color patch. There was also no visual change in surface sheen in the image areas. However, further research is suggested to assess this technique, including investigation of dye migration into the paper substrate.
Inkjet prints created on Ilford Galerie Gold Fibre Silk paper should not be cleaned using immersion techniques. The cracks that formed during this study may have in part been due to the drying method used, however full immersion is unadvisable until further research can be conducted. The results of immersion of the Epson Ultra Premium Photo Paper Luster suggest that this may be a possible cleaning technique for this paper if the print can be dried in a manner that tidelines are unable to form.
Thanks to Alumni Relations at the University of Delaware for providing two Alumni Enrichment Awards, one of which provided funding for a 2008 summer internship and another which allowed the author to attend the PMG Winter Meeting in Tucson, Arizona to present this research. The author would also like to thank the Undergraduate Research Program for providing an Undergraduate Research Summer Fellowship, which also contributed to funding the 2008 summer internship at Aardenburg Imaging and Archives.
The author would especially like to thank Jae Gutierrez for assistance and advice throughout the project, as well as Mark McCormick-Goodhart for providing the internship that made this project possible. Special thanks to all of those who assisted in my research and in writing the thesis, including Debbie Hess Norris, Leslie Reidel, Dick Sacher at IT services, Oleg Baburin of Chicago Albumen Works, Martin Jurgens, Dr. Joe Weber, and Meg Meiman.
TESSA GADOMSKI
University of Delaware
Arney, J.S. et al. “Analysis of Paper Gloss.” Journal of Pulp and Paper Science 32, no. 1 (2006): 19-23.
Arney, J.S. and David Nilosek. “Analysis of Print Gloss with a Calibrated Microgoniophotometer.” Journal of Imaging Science and Technology 51, no. 6. (2007): 509-513.
Arney, J.S. and Ling Ye. “Gloss Granularity of Electrophotographic Prints.” Journal of Imaging Science and Technology 51, no. 4 (2007): 293-298.
Arney, J. “Technique for analysis of surface topography of photographic prints by spatial analysis of first surface reflectance.” The Journal of Imaging Science and Technology 46, no. 4 (2002): 350-358.
Berns, Roy S. Billymeyer and Saltzman’s Principles of Color Technology Third Edition. New York: John Wiley & Sons, Inc. 2000.
ColorChecker Colorimetric Data. X-Rite, Incorporated and GretagMacbeth AG/LLC USA. 2008. November 2008. http://usa.gretagmacbethstore.com/index.cfm/act/Catalog.cfm/catalogid/1742/c
Daniels, V. and J. Kosek. “Studies on the Washing of Paper: Part 2: A Comparison of different washing techniques used on an artificially discoloured, sized paper.” Restaurator, 25. (2004): 260-266.
Epson Stylus Pro 4800-Print Permanence Ratings (preliminary). Wilhelm Imaging and Research. 2005. http://www.wilhelm-imaging.com.
Ilford Galerie Gold Fibre Silk. Ilford. February 2009. http://www.ilford.com/en/products/galerie/gold/goldMediaFibre.asp.
Harold, Richard W. and Richard S. Hunter. The Measurement of Appearance, Second Edition. New York: John Wiley & Sons. 1987.
Material Safety Data Sheets/PIS. Epson. 2000-2009. February 2009. http://www.epson.com/cgi-bin/Store/support/Supportmsdsmain.jsp.
McCormick-Goodhart, Mark and Henry Wilhelm. “The Correlation of line quality degradation with color changes in inkjet prints exposed to high relative humidity.” In IS & T’s NIP19: 2003 International Conference on Digital Printing Technologies. 2003. 420-425.
Messier, Paul and Timothy Vitale. “Effects of Aqueous Treatment on Albumen Photographs.” Journal of the American Institute for Conservation. 33. (1994): 257-78. http://albumen.stanford.edu/library/c20/messier1994.html.
Morrison, Pip. Solvent Effects on Silver Dye Bleach Materials. George Eastman House & Image Permanence Institute. July 11, 2005. http://www.arp-geh.org/indexsep.aspx?nodeidp=203.
Papers presented in Topics in Photographic Preservation, Volume Thirteen have not undergone a formal process of peer review.