
Topics in Photographic Preservation 2011, Volume 14, Article 16 (pp. 93-102)
Presented at the 2011 PMG Winter Meeting in Ottawa, Canada
In 2005 the Canadian Conservation Institute (CCI) received a request from the Medicine Hat Museum and Art Gallery (now part of the Esplanade Arts & Heritage Centre) in Medicine Hat, Alberta, for the treatment of a large silver gelatin panoramic photograph of the city of Medicine Hat. It clearly met the criteria for a CCI treatment project based on its strong historical significance to the community and the rarity of the large format. With the imminent arrival of a photograph conservation intern from the Winterthur/University of Delaware Program in Art Conservation, the piece was called in for treatment, arriving in the fall of 2008. The photograph was produced in July 1912 for the young city of Medicine Hat by L. C. Barton of Chicago. It measures 21.7” x 145.7” and is printed on a single sheet of photographic paper.
This paper is a personal account of the treatment of the panorama including, the problems encountered during treatment, the thought processes and rational when confronting the problems and the eventual solutions.
Medicine Hat of the early 20th century, with its extensive natural resources, thriving industry, and rapidly growing community, was known as “the Pittsburgh of the West” - a positive appellation at the time. The panorama displays the entirety of the young city; the hotels, homes, schools, hospitals, and factories which grew into the industrialized Medicine Hat of the present-day.

Figure 1: the Medicine Hat Panorama, before treatment
© All rights reserved. Reproduced with the permission of the Canadian Conservation Institute of the Department of Canadian Heritage, 2009
Shortly after the introduction of photography an interest in panoramic photographic images developed, with the first known patent for a panorama camera granted in 1843 in Germany. Before the development of cameras that exposed wide horizontal images onto plates and later onto film, the panorama effect was accomplished by placing sequential, individually photographed and processed prints or plates next to each other in the appropriate order. With the introduction of flexible film bases in the late 19th century, panoramic photography was ubiquitous. Many dedicated panoramic film cameras were on the market; the Cirkut camera being the most well known in North America. These cameras involved a rotating lens and stationary film or a rotating lens and rotating film. The resultant single large negative was then contact printed onto one sheet of photographic paper (Lenman 2005).
The Medicine Hat Panorama, though dating to a time when the rotating-component panoramic cameras were in use, employs the earlier individual sequential negative technique. Contact printed on one continuous sheet of silver gelatin photographic paper, the Medicine Hat Panorama was produced using six separate large format negatives (each 55 cm high and 68 cm wide). Each negative exposed and processed individually was then placed in the proper alignment on the light-sensitive photographic paper and then exposed to light, creating a contact print. The evenness of the exposure and the smooth integration of one plate into another is a testament to the great skills of the photographer and printer.
The panorama entered the collection of the Medicine Hat Museum in 1962 in less-than-optimal condition. When it arrived at CCI, the panorama was attached to a sheet of plywood and prior to this backing had been roughly torn into three segments. There were also numerous other cracks and losses and the surface was covered in a heavy layer of dirt and grime. At some point in the past attempts were made to repair the damage and to reduce the surface soiling. Unfortunately these good intentions left behind areas of heavy abrasion, extensive disfiguring retouching, and multiple damaging adhesive residues.

Figure 2: the Medicine Hat Panorama - the locations of major damage
--- major tears
 major loss
major loss
 severe surface abrasion
severe surface abrasion
 major cracks in emulsion
major cracks in emulsion
© All rights reserved. Reproduced with the permission of the Canadian Conservation Institute of the Department of Canadian Heritage, 2009
We began this project knowing that we would face multiple challenges: a photograph adhered overall to a sheet of plywood; a heavily soiled surface of unknown stability; and a massive amount of inpainting necessary to reintegrate the image. These difficulties were compounded by the twelve foot length of the piece. The plan was to control the risks and minimize uncertainties as much as possible. Where applicable, this involved both scientific analysis and extensive spot testing prior to embarking on each phase of the treatment.
The first challenges were easily met. Several adhesive samples from the Medicine Hat panorama were analyzed by CCI’s Analytical Research Laboratory using Fourier transform infrared (FTIR) spectroscopy (Helwig 2008). This was done in order to characterize the several unknown adhesives present on the photograph. Identification of the various adhesives was invaluable in identifying which solvents would be effective in reducing/removing the adhesives.
After the adhesive residues were addressed, the recto of the photograph was dry cleaned with vulcanized rubber sponges. Some old and disfiguring retouching in the form of graphite was removed in addition to loose surface grime.
The ninhydrin stain for primary and secondary amines was used to help select the optimal aqueous surface cleaning solution (Wang 2009). The ideal cleaning solution would provide maximum cleaning power with minimum surface disruption. Testing was carried out on both a silver gelatin photograph of approximately the same age and condition as the Medicine Hat panorama (part of the CCI study collection) and the panorama itself. Both photographs were also sampled for analysis using scanning electron microscopy (SEM).
After the analysis, the cleaning solutions were evaluated and the print was surface cleaned with two different PhotoFlo and water solutions. The majority of the surface was cleaned using a 0.1% PhotoFlo 200 in water solution. The results of the testing utilizing the ninhydrin stain for proteins indicated that the 0.1% PhotoFlo 200 in water solution disturbed the gelatin the least and the back scattered electron imaging in the SEM also indicated that this solution was the least disturbing to the gelatin binder (Sirois and Bailey 2009). A higher concentration, 0.5% PhotoFlo in water was used in areas of more tenacious soiling. Large swabs and wads of cotton were used to apply the aqueous cleaning solutions and remove the surface soiling. Care was taken to ensure that the cleaning was as even as possible.
We anticipated that our most difficult task would be to remove the photograph from the plywood tertiary support. Small-scale solvent testing indicated that both xylene and toluene solubilized small amounts, and softened large bulky masses, of the adhesive used to attach the object to the plywood. This was encouraging, but options for unmounting were still constrained because of the physical nature of plywood and because of the unwieldy length of the object (twelve feet).
The emulsion was quite stable during the aqueous surface cleaning, but it was decided to consolidate in preparation for removal from the plywood anyway. A 1% gelatin solution (275 bloom) in filtered water was lightly sprayed onto the surface of the photograph and dried with the aid of a hairdryer. The hairdryer was used to limit the possible formation of tidelines. This process was repeated five times.
The object was removed from the plywood backing by releasing the adhesive (a synthetic rubber contact cement) between the secondary support and the plywood. A syringe was used to introduce xylene into the adhesive interface between the secondary support and the plywood. Thin spatulas were then used to assist in the separation process. Because of the solvent use, this procedure was carried out within a spray booth and using respirators.

Figure 3: removal from the plywood tertiary support
© All rights reserved. Reproduced with the permission of the Canadian Conservation Institute of the Department of Canadian Heritage, 2009
As the photograph was removed from the plywood, it was rolled up onto a specially-constructed large-diameter tube. This allowed the separated portion of the panorama to be kept safely out of our working space and enabled safer and easier access to the adhesive layer. The tube was constructed from a plastic well liner (purchased at Home Depot) that was lightly padded and then covered with glassine paper.
After separating all of the components, reducing adhesive residues, cleaning the tear edges, and reducing creases, the loose fragments were reattached, the tears were mended, and the large losses filled with toned paper.
Spirits were high: the project was ahead of schedule and everything was proceeding smoothly and as expected. Most importantly, we believed that the most challenging stages of the treatment were over. Our careful preparation had been rewarded.
The panorama had two major tears, structurally compromising the entire object: we felt that it was necessary for the overall stability of the piece, for it to be lined. Therefore, it was lined with 4 ½ pieces of a heavy-weight Japanese paper and wheat starch paste.
Unfortunately, during the lining process, moisture migrated through the paper support to the emulsion, staining the recto of the image. Although we were alarmed, due to the previous experience with aqueous treatments that had been carried out on this piece, we thought that when thoroughly dry, the water marks would disappear. This was not to be.
Our first stain removal attempt involved spraying the stained areas with a 1:1 water/ethanol solution followed by blotting the surface, hoping to at least disperse or diffuse the stain. This technique proved ineffectual and was followed by several attempts at localized stain reduction using a variety of suction platens. Stained areas were wet with filtered water and ethanol: an approximately 90% ethanol in water solution was also used as a transitional barrier and to decrease the potential for tidelines. This also proved ineffectual, but did not exacerbate the initial staining problem.

Figure 4: post-lining stains
A second attempt at solubilizing or at least diffusing the stain was to immerse the photograph in a bath of filtered water and alcohol. An initial test was carried out on one end of the photograph: approximately six inches of the photograph would be immersed into a 10% ethanol water bath. Prior to the test, the Japanese paper lining was removed from the portion of the photograph to be immersed.
The test portion of the print was wet with water and alcohol (10% ethanol in water). As before, an approximately 90% ethanol in water solution was used as a transitional barrier between wet and dry, decreasing the potential for tidelines.
The six-inch test portion of the photograph was immersed and after approximately 10 seconds it was clear that the bond between the gelatin emulsion and the baryta layer was compromised. The emulsion began to solubilize, primarily in the sky region. The photograph was immediately removed from the bath and placed on the suction table with no loss of image but with some reticulation of the gelatin. The surface was lightly sprayed with water and ethanol in an effort to limit the development of potential tide lines. Although some staining material was transferred to the blotter, there was little reduction of the stain on the recto. Regardless, the fragility of the emulsion layer precluded further attempts at immersion.
The reaction of the emulsion to immersion was highly unexpected, as it had been stable during all of the previous treatment steps. It was previously hypothesized, due to the streaky and heavily abraded sky areas, that harsh cleaning methods (primarily rough scrubbing) had been used in the past to attempt to make the sky light. In light of the reaction to immersion, we hypothesize that these past cleaning attempts may have involved the use of strong cleaning solutions, possibly highly alkaline. These would have detrimentally affected the gelatin emulsion though not in a manner that would be discernable during visible examination. The bond between the gelatin emulsion and the baryta layer is what failed during the immersion. It is also possible that there was an inherent instability in the structure of the photographic paper when manufactured that could later cause the gelatin-baryta bond to fail.
The question remained, how to get rid of the staining that was so evident primarily in the sky region of the image, ostensibly a non image area.
The one remaining option was the localized application of a bleach. Two sodium borohydride solutions were prepared at, 1% in water, and 1% in ethanol. Though the sodium borohydride effectively reduce the staining, as a reducing bleach it also reduced remaining silver salts found in the emulsion. This resulted in a darkened of the test spots to a light grey tone which was deemed unacceptable.
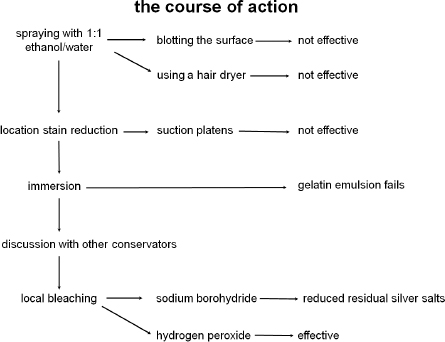
Figure 5: decisions leading to local hydrogen peroxide bleaching
Various concentrations of hydrogen peroxide in water were then tested. Multiple light applications of 8% hydrogen peroxide in water were effective in significantly reducing the appearance of the stains. However, as a strong oxidizing agent for image silver it was viewed with great suspicion and many spot tests carried out across the image silver. Due to its highly uniform image tone and complete lack of image staining, the assumption was that it had been toned making it less susceptible to oxidation. The results of all of the spot tests were negative and the decision was made to proceed with the treatment which included localized applications of an 8% hydrogen peroxide solution applied with a fine brush followed by rinsing. Some of the very larger areas were masked off and the solution applied as a light mist. All areas to which the bleach had been applied were then rinsed multiple times with pure water, repeatedly sprayed and blotted in an attempt to remove as much residual peroxide as possible. This cycle of bleach application, water wetting, and blotting was repeated as necessary to reduce the appearance of the stains. Prior to treatment, other conservators were consulted and the protocol refined.

Figure 7: image staining, left half of the panorama
© All rights reserved. Reproduced with the permission of the Canadian Conservation Institute of the Department of Canadian Heritage, 2009
As we reached approximately the ¾ completion mark with successful stain reduction throughout and no apparent impact on the image silver, things changed. Stain removal was completed on an area in the river, not the first staining in the river area to be bleached, however the first to react.
Gradually staining of the image began to appear and proceeded quickly in the image area close to where the bleaching had taken place. Repeated rinsing and blotting did little to slow down the development of the stain. However, by the time we left for the weekend, it being Friday, we felt confident that we had arrested the oxidative process. However, nothing could have prepared us for the extent of the discoloration that confronted us on Monday morning. Though the size of the affected area had not increased, the intensity had.
Intense consultations, begun the previous Friday with CCI scientists, and photochemists intensified. Eventually they agreed that our suggestion to attempt to reduce the stained image silver with a reducing agent such as sodium borohydride was a reasonable approach. A 2% sodium dithionate solution, a stronger reducing agent than sodium borohydride, was also recommended and tested however it proved less effective than the borohydride and it was abandoned.
The function of the reducing bleach was twofold, to reduce the oxidized silver back to a metallic state and to react with and consume any remaining hydrogen peroxide within the object.
The stained area was carefully, repeatedly (obsessively) rinsed and blotted with filtered water in an attempt to remove/dilute the bleach. A dilute solution (approximately 0.1%) of the reducing bleach sodium borohydride was tested on the oxidized areas and was found to be effective at increasing the image density and shifting the image color back to an appropriate tone, more in the dark purple range. The application of the sodium borohydride thus continued with concentrations ranging from approximately 0.1% to 0.3% in solutions of water, 1:1 water/ethanol, and ethanol.
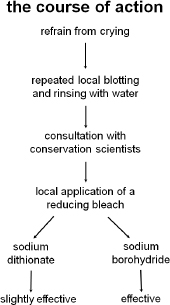
Figure 6: decisions leading to sodium borohydride application

Figure 8: before (left) and after (right) sodium borohydride application, losses were filled, and with most of the inpainting complete
As we now profoundly understand, hydrogen peroxide can linger in substrates for upwards of four/five days, this fact accounted for the reversion of the affected areas back to orange following borohydride treatment, necessitating numerous applications of the borohydride in various spots. The borohydride treatment was also followed by thorough water and alcohol rinse. Finally it ceased to react and we proceeded to the next steps in the completion of the treatment including a second round of consolidation and relining the end of the print that had been removed for immersion, as well as infilling, retouching, hinging and mounting.
Although the sodium borohydride applications were successful in restoring much of the image density, what they did not restore was the original image tonality. This is because when the oxidized silver was reduced, the resulting silver particles were of a different size and/or shape than the original silver particles. As a result of this, the areas of damaged image silver were noticeably warmer in tonality that the original undamaged photograph. This was visually disturbing and required some unconventional inpainting: translucent glazes of varying shades of peach.
Applying a transparent light color over a dark color will result in a color that is cooler than intended. Because of this effect, a glaze that matched the original tonality, when applied to the over-warm image areas, would appear to be quite blue. A warmer-than-expected glaze was necessary to counteract this effect. After inpainting, the damaged area is still slightly warmer in tonality, but is not eye-catching.
Since most of the tears also involved losses to the primary support along the tear edges, the gaps were filled with a mixture of barium sulfate and gelatin, applied with a brush. All of the inpainting was done with watercolors.

Figure 9: the Medicine Hat Panorama, after treatment
© All rights reserved. Reproduced with the permission of the Canadian Conservation Institute of the Department of Canadian Heritage, 2009
In spite of our best efforts to understand the stability of objects, whether they be photographs, works of art on paper or sculptures, we place them at risk everytime we touch them. Anyone who has been in the conservation field for a while understands this. Mitigating risk in essence becomes our primary responsibility. Multiple spot tests and the extraordinary investigative lengths we go to as professionals in order to inform ourselves about the properties of the object are essential steps in every treatment and yet they tell only part of the object’s story. One must exercise vigilance in every aspect of a treatment. Though students and teachers alike must assume responsibility for vigilance at every turn, it ultimately rests on the shoulders of the supervising conservator In the case of the Medicine Hat panorama, the supervising conservator should have been more vigilant throughout the treatment. Staining did occur and this ultimately became a story of pursuing options for recovery. That pursuit was necessarily exhaustive and inclusive. Many colleagues were consulted and ultimately much was learned
Catharsis aside, we, the conservators on this project, present this paper to inform and encourage others to talk about “problematic treatments”. If the old adages are true, that “we learn from our mistakes” and “experience is the best teacher,” and they surely are, we all benefit from disclosure.
Helwig, K. “Analysis of samples from a panoramic photograph of the city of Medicine Hat for Medicine Hat Museum and Art Gallery, Medicine Hat, Alberta, Canada.” CCI Report No.ARL 4597: 1 October 2008.
Lenman, R. The Oxford Companion to the Photograph. Cambridge: Oxford University Press, 2005.
Sirois, J, and K. Bailey. “Analysis of a panorama photograph of the city of Medicine Hat for Medicine Hat Museum and Art Gallery, Medicine Hat, Alberta, Canada.” CCI Report No.ARL 4634: 24 April 2009.
Wang, N. S. “Amino acid assay by ninhydrin colorimetric method.” Department of Chemical & Biomolecular Engineering, University of Maryland. Accessed 4 August 2009. http://www.eng.umd.edu/~nsw/ench485/lab3a.htm
GREG HILL
Canadian Conservation Institute
JESSICA KEISTER
Conservation Center for Art & Historic Artifacts
Papers presented in Topics in Photographic Preservation, Volume Fourteen have not undergone a formal process of peer review.