Topics in Photographic Preservation 2011, Volume 14, Article 39 (pp. 237-249)
The purpose of this experiment was to determine whether the belief that cyanotype prints will be adversely affected by buffered enclosures in long-term storage is truly valid. This assumption has been supported by anecdotal reports, experiments with alkalis on Prussian Blue, and experiments with calcium carbonate paste directly on cyanotype prints. The effect is not known to have been established experimentally using actual, commercial buffered and non-buffered storage papers (interleave tissues, envelope papers, folder stocks etc.). Also, contrary to the above, it has been theorized that since calcium carbonate buffering is in a mostly solid form in paper (it has a low solubility and the moisture content of paper is typically very low), it is not likely to be reactive with the cyanotype colorants (or other imaging colorants such as chromogenic dyes or binders such as albumen). In fact, ISO 18902 Imaging materials -- Processed imaging materials -- Albums, framing and storage materials now recommends the use of buffered papers for storage of all photographic materials; however, cyanotypes are not specifically discussed. Archival suppliers would prefer to stock only buffered papers as they are more widely used. In addition, it is becoming more difficult for suppliers to find paper mills willing to produce high-quality non-buffered papers. It would also make it easier on institutions if one enclosure paper could be used for most if not all print types. These contradictions between theories, experience, experimental results, and standardized recommendations need to be rectified. In these new experiments, papers of varying pH as well as commercial buffered and non-buffered papers were artificially aged in contact with prints created using several cyanotype formulations. It was found that some cyanotype formulations were more sensitive to damage than others, but all were sensitive to both alkaline and neutral papers. Only acidic papers fully protected the images.
The purpose of this experiment was to determine whether the belief that cyanotype prints will be faded or yellowed by buffered enclosures in long-term storage is truly valid. This belief has been supported by anecdotal reports, experiments with alkalis on Prussian Blue (Holtzman 1945), and experiments with calcium carbonate paste directly on cyanotype prints (Ware 1999). The effect is not known to have been established experimentally using actual, commercial buffered and non-buffered storage papers (interleave tissues, envelope papers, folder stocks etc.). Ware demonstrated a potential reactivity in an experiment by applying a paste made with distilled water and calcium carbonate directly to the surface of cyanotype prints and exposing them to high humidity for forty-eight hours. Fade of the image was reported, but Ware stated that the method may have been unrealistic and that the results should be taken with some caution (Ware 1999). Contrary to all of the above, it has been theorized that since calcium carbonate buffering is in a mostly solid form in paper (it has a low solubility and the moisture content of paper is typically very low), it is not likely to be reactive with the cyanotype colorants (or other imaging colorants such as chromogenic dyes or binders such as albumen). In fact, ISO 18902 Imaging materials -- Processed imaging materials -- Albums, framing and storage materials now recommends the use of buffered papers for storage of all photographic materials; however, cyanotypes are not specifically discussed (ISO 18902:2007). In addition, archival supply companies would prefer to stock only buffered paper as it is more widely used, and it is becoming more difficult to find paper mills willing to produce high quality non-buffered papers. It would also make it easier on institutions if one enclosure paper could be used for most if not all print types. These contradictions between theories, experience, experimental results, and standardized recommendations need to be rectified. In these experiments, prints made with three different cyanotype formulas were artificially aged in contact with papers of varying degrees of pH. The results were then used to determine whether buffered or non-buffered paper enclosures can be recommended.
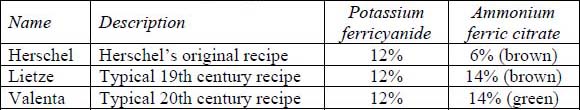
The cyanotype prints were created by coating ammonium iron(III) citrate (brown or green) and potassium ferricyanide onto Buxton paper (made by Ruscombe Paper Mills in France) by dragging the solution across the paper’s surface using a glass rod. The glass rod allowed for a more even coating than application by brush. Three different cyanotype formulas were tested individually against five papers of varying pH for a total of fifteen different combinations. The cyanotype formulas (Ware 1999) are shown in table 1:
Table 1. Cyanotype Test Print Formulations

According to Ruscombe Paper Mills, the Buxton paper should be “chemically compatible with iron-salt-based sensitizers and be archivally sound. The white paper is produced from 100% security grade cotton fiber, peroxide bleached with no optical brightening agents or alkaline buffering agents such as chalk (calcium carbonate). The cotton is beaten to a high bonding strength, with only a neutral reactive internal sizing agent [alkyl ketene dimer] to ensure a good degree of impermeability. The handmade sheets are heavily pressed several times in both wet and dry states to reduce porosity and to create the desired surface. The surface is lightly textured due to the impact of the felt mark derived from the woven woolen felts which are used.” (Ruscombe 2011).
After the coated papers had dried, they were placed with 10-step transmittance wedges into glass-covered contact printing frames to create images of varying levels of tone. They were then exposed to UV light (artificial) for one hour to create the images. The paper was then rinsed in tap water for 30 minutes and hung until completely dry. They were then cut into 13x2 cm strips in order to fit the metal test jigs used during artificial aging.
Two types of test papers were used with varying pH. First were papers of high and low pH using a highly soluble, strong acid or base to act as worst-case scenarios. For these, strips of Whatman No. 1 filter paper, cut to 13x2 cm, were soaked in 0.1 N hydrochloric acid (HCl) or 0.1 N sodium hydroxide (NaOH) for 10 seconds. They were then dried for 24 hours laid flat on sheets of polyester. Polyester was used as opposed to blotter paper to prevent the leaching out of the acid or alkaline solution. Strips of untreated filter paper were also evaluated.
In addition, commercial non-buffered and buffered papers were also tested. These included 145# Non-Buffered Photographic Folder Paper and 145# Lig-free Type I Folder Stock (buffered 3%). Both were purchased from Conservation Resources International in Springfield, VA.
The pH of all of the test papers was measured to quantify their levels of acidity or alkalinity. To do this, 1 gram of each material was placed into 100-ml glass beakers. Then 75 ml of ASTM D 1193 (Types I-IV) reagent grade water with a pH between 6.5 and 7.5 was added to each beaker, ensuring that the paper was completely soaked. The beakers were then covered with Parafilm (to prevent absorption of CO2 from the air) and left sitting for one hour. After one hour, a Mettler Toledo SevenEasy pH meter with VWR Symphony gel epoxy combination electrode was calibrated and used to make measurements of each sample. Tests were performed in duplicate and the results averaged. Table 2 shows the pH results for all of the test papers.
Table 2. pH of Test Papers
The method described in ISO 18916 Photographic Activity Test (PAT), was modified to test the cyanotype prints against the test papers (ISO 18916:2007). The standard method normally involves the accelerated aging of special image interaction and gelatin stain detectors against the enclosure materials being studied. The reactivity of the enclosure material on the detectors is then compared to the reactivity of a known “good” control (Whatman No. 1 filter paper). Limits to differences in optical density changes between the two determine whether the enclosure material passes or fails the test. In the standard test, the image interaction detector is a thin colloidal silver/gelatin layer on a polyester base and is used to screen for reactions that cause image oxidation or reduction. The gelatin stain detector is a photographic paper processed to minimum density (paper white) and is used to screen for reactions that cause print yellowing. For this experiment, strips of cyanotype prints replaced the standard detectors. These detectors were created with multiple levels of density to evaluate both image interaction and stain in one strip.
All of the cyanotype test strips were evaluated both quantitatively and qualitatively. The cyanotype strips were read both before and after artificial aging with a GretagMacbeth Spectrolino/Spectroscan spectrodensitometer for ANSI Status A red, green, blue, and visual densities as well as CIELab values with CIE ΔE (1976) calculated. All of these values were collected because it was unknown at the outset of the experiment which measurement would be most useful. The strips were also visually assessed after artificial aging for signs of fade, color shift, paper yellowing, or any other forms of damage.
The jigs were then assembled according to the ISO 18916 standard with each jig containing two replicates of one type of cyanotype print (e.g. Herschel) and one test paper (e.g. commercial non-buffered paper). The filter paper interleave required by the standard was not used in this test to maximize contact between test papers and the cyanotype prints. Fig. 1 shows the assembly order of the cyanotype prints and test papers in the jigs. The loaded jigs were then placed in an Espec model LHL-112 temperature and humidity cabinet at 70°C and 86% RH for 15 days. After this time, the jigs were removed from the oven and the cyanotype detectors read again with the spectrodensitometer to determine if any change occurred.

Figure 1. Jig Assembly Diagram
In addition to the above experiment, a repeat of Ware’s experiment using a worst-case test of calcium carbonate directly on the surface of a cyanotype print was repeated (Ware 1999). A paste of calcium carbonate and distilled water was spread over the surface of strips of cyanotype prints made using the three different formulations. These were placed in an Espec model LHL-112 temperature and humidity cabinet at 21°C and 95% RH for 48 hours. After the incubation the CaCO3 was brushed off as much as possible and then the samples were measured and visually assessed for damage.
All of the cyanotype prints were damaged significantly (both fade and conversion of image to yellow) when incubated in contact with the NaOH-treated paper, the untreated filter paper, and both the buffered and non-buffered papers. The prints in contact with the HCl-treated filter paper retained image quality.
Table 3 shows images of the Herschel formula detectors for each test paper type. The print in contact with the HCl-treated paper did not appear to change. All of the other prints faded and turned yellow across the entire tonal range. The most significant amount of fade occurred in the print in contact with the NaOH-treated paper. The untreated filter paper, buffered and non-buffered papers caused roughly equivalent levels of damage to each other.
Table 3. Detectors in Contact with Herschel Prints
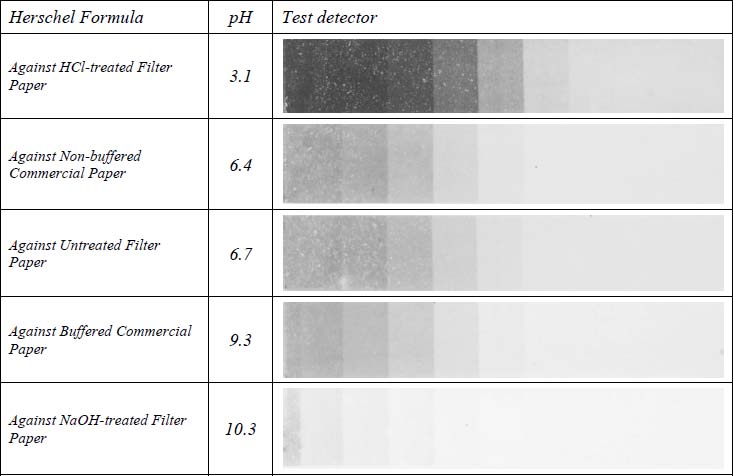
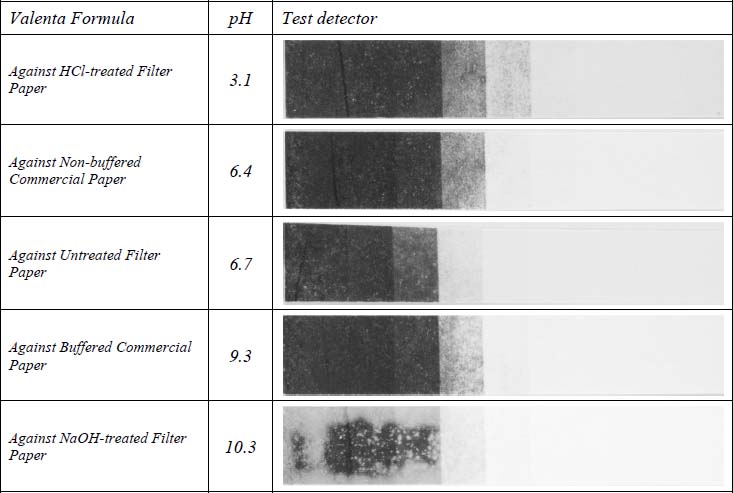
Table 4 shows images of the Lietze formula detectors for each test paper type. The strip in contact with the NaOH-treated paper showed uneven fade across the highest initial density levels with the edges fading more than the center. Areas that faded were converted to yellow as with the Herschel prints. The untreated filter paper, buffered and non-buffered papers caused roughly equivalent levels of damage to each other. There was conversion of the image to yellow in the low to mid-tones but much of the image remained blue in the darkest areas. The highest density step did show some yellowing due to its proximity to the edge of the print. The print in contact with the HCl-treated paper did not appear to change at all.
Table 4. Detectors in Contact with Lietze Prints

Table 5 shows images of the Valenta formula detectors for each test paper type. As with the Lietze formula samples, the Valenta strips in contact with the NaOH-treated paper showed uneven fade across the highest initial density levels with the edges fading more than the center. Areas that faded were converted to yellow as with the Herschel prints. The untreated filter paper, buffered and non-buffered papers caused similar but not identical damage and could be ranked from best to worst: non-buffered, buffered, and untreated filter paper. The low to mid-tones were converted to yellow, but the image remained blue in the darkest areas. The highest density step did show some yellowing due to its proximity to the edge of the print but less so than the Lietze prints. The print in contact with the HCl-treated paper did not appear to change at all.
Table 5. Detectors in Contact With Valenta Prints
Figures 2 and 3 below show the effect of the damage on the tonal range of the image. The Herschel formula print in contact with the HCl-treated paper showed only a very slight increase in image density (fig. 2). This matches the visual assessment and applies to all three cyanotype formulations in contact with the HCl-treated paper. The increase in density may be attributable to thermal yellowing of the paper support. The Herschel print in contact with the NaOH-treated paper lost nearly all of its image density at every step (fig. 3).
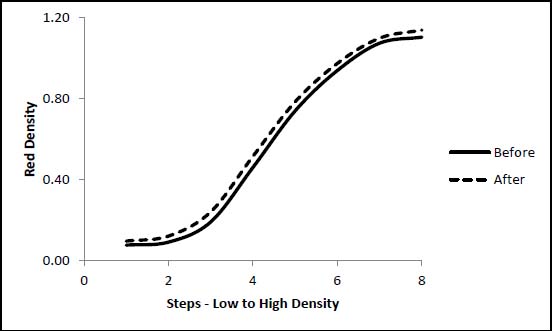
Figure 2. Effect of HCl-treated Paper on Herschel Formula Print Tonal Range
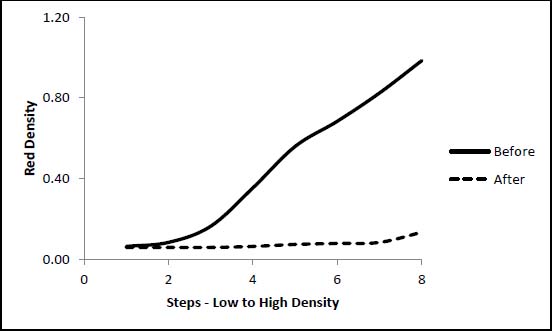
Figure 3. Effect of NaOH-treated Paper on Herschel Formula Print Tonal Range
The total loss of density across the tonal range in the NaOH-treated sample belies the fact that some of the image was converted to yellow which Status A Red density, would not have picked up. Figure 4 shows how even after almost all of the blue color is lost to fade (red density), a significant level of image is formed in yellow (blue density).
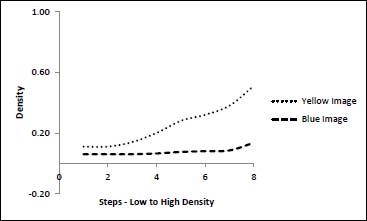
Figure 4. Comparison of Red and Blue Densities in NaOH-faded Herschel Print
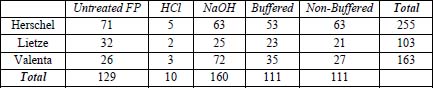
Because the prints did not simply fade but also converted to yellow at some or all tone levels, ΔE was ultimately considered the best measure as it incorporates both fade and hue shift. Table 6 shows the highest ΔE for each cyanotype formulation and test paper combination. In an attempt to quantify the overall sensitivities of each cyanotype formulation as well as the reactivity of each test paper type, the columns and rows were summed to create a total score for that formulation or test paper. It shows that the Herschel formula was most prone to damage followed by Valenta and Lietze. It also shows that the NaOH-treated paper was most reactive followed by the untreated filter paper. The buffered and non-buffered papers were equivalent while the HCl-treated paper caused almost no change at all.
Table 6. Highest ΔE on Each Step Wedge
The numerical results matched the visual assessments for the HCl-treated samples but not the others. When test prints are severely degraded, numerical measurements tend to vary to a higher degree. This applies to all permanence studies and not just to cyanotypes. Rankings between treatments on small variations are therefore potentially problematic, especially with small numbers of test samples as in these experiments. The mottling appearance of some prints, such as the Lietze print in contact with NaOH treated filter paper (fig. 5), as well as the exaggerated fade on the highest density steps due to their proximity to the edge of the paper all increased the variability of the measurements.

Figure 5. Mottled Detector – Dark Areas Are Cyan and Light Areas Yellow
The differences between the ΔE values for the NaOH-treated, buffered, non-buffered, and untreated filter paper samples are not nearly as significant as the differences between all of these and the HCl-treated sample. Using the data, there is not necessarily a clear choice among the NaOH-treated, buffered, non-buffered, and untreated filter paper samples upon which to make a recommendation. The visual analysis of the samples provided a much clearer picture as the cyanotypes in contact with the NaOH-treated paper appeared dramatically more faded than the others. While the NaOH-treated samples gave an idea of what a highly alkaline sample might do, it is not a realistic threat as enclosures rarely contain such strong bases in their formulations (though starch adhesives used in mass manufacturing of mat and corrugated boards sometimes do).
Tables 7-9 shows the ΔE for each cyanotype formulation and test paper combination for paper white areas (Dmin), mid-tones (Dmid) and maximum initial density (Dmax). The largest change did not always occur at the point of initial highest density. In an attempt to quantify the overall sensitivities of each initial density level, the columns were summed to create a total score for that density level for each paper formulation. The lightest print areas changed very little and may be due more to thermal yellowing of the paper than loss or conversion of image material. On average, the Herschel and Valenta formulation prints were most sensitive in the highest densities while the Lietze prints were most sensitive in the mid-tones. This did not apply to every specific print formulation and test paper combination (e.g. Herschel print in contact with buffered commercial paper).
Tables 7. ΔE for Various Initial Density Levels for Herschel Prints
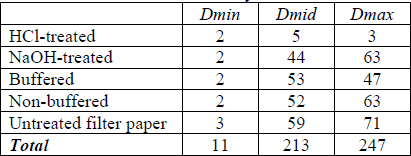
Tables 8. ΔE for Various Initial Density Levels for Lietze Prints
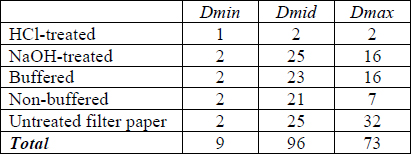
Tables 9. ΔE for Various Initial Density Levels for Valenta Prints
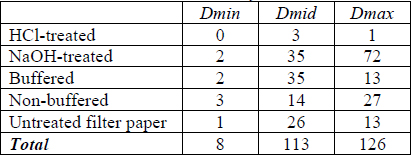
The prints in the repeat of the Ware experiment faded but did not convert to yellow. This was consistent with Ware’s results (Ware 1999). Table 10 shows the highest ΔE for each cyanotype formulation and test paper combination. It was difficult to measure changes in lightness due to residual CaCO3 on the surface of the print, which could not be entirely removed.
Table 10. Highest ΔE for Each Print Type in Repeat of Ware’s Experiment

Ware stated that “Calcium carbonate only poses a clear threat to cyanotypes when in direct contact with the image; it has little ability to migrate through cellulose, so the dangers of chalk-buffered enclosures may have been somewhat overstated” (Ware 1999, 131). Before this current investigation, the authors of this paper would have agreed with the above statement; however, given the results of these experiments we now believe that assumption should be questioned.
While it was expected that the NaOH-treated and possibly the buffered paper would result in deterioration of the cyanotypes, it was unexpected that the non-buffered and untreated filter papers would also result is such significant damage. For alkaline hydrolysis of the image to occur there must be a source of OH-. The source is clear with NaOH, and the rapid fade of the image expected. CaCO3 in water tends to indirectly create a surplus of OH- ions; however, to a lesser concentration than the direct addition of OH- from dissolved NaOH. Also, CaCO3 has a low solubility, so most of it remains in a solid form. For the neutral materials (the non-buffered and untreated filter papers) it was not clear where the OH- was coming from. It was possible (but considered unlikely) that it was coming from the Buxton or Whatman papers themselves, so these were tested for pH as well (table 11.).
Table 11. pH of Whatman and Buxton Papers

The pH of the Whatman and Buxton papers matched that of the test water indicating that they are pH neutral and should not contribute to alkaline hydrolysis of the cyanotype image. This brings the question: why did the cyanotypes in this experiment fade in the absence of alkali? It is possible that all Fe(III) is unstable in non-acidic conditions (including neutral), and this is the cause of the fade (and potentially even the conversion to yellow) of the cyanotype image in these experiments (Flynn 1984). Also, why have the cyanotypes already in collections not faded in contact with non-buffered papers? It is theoretically feasible that acidic papers used in period cyanotypes or their mounts have provided some degree of protection for these prints over the years. Further study is recommended.
The following conclusions were drawn from the data:
The experiments clearly showed that alkaline enclosures should not be used. However, the data also precludes making a recommendation for neutral pH enclosures as they also resulted in significant damage to the test cyanotypes especially the most sensitive formulation. On the other hand, it cannot be recommended that cyanotypes be stored in acidic enclosures as they may deteriorate the paper substrates.
It would be helpful to know just what levels of acidity actually create the protective effect seen in these experiments. If it is not too far from neutral, it may be possible to balance image permanence with paper permanence for maximum object longevity. It would be also helpful to know if alkaline enclosures must be in direct contact with cyanotypes for the reactions to occur (i.e. can buffered boxes be used). Finally, it would be useful to know whether the use of plastic enclosures or interleaves such as polyester would address the issue. Plastics were not tested and could be a potential replacement for paper enclosures; however, as Ware pointed out this would be problematic for images exposed to light when inside the enclosure because plastic does not allow for sufficient air exchange (Ware 2003). Still, they may potentially be valuable for dark storage of cyanotypes.
The authors would like to thank Dr. Michael Ware for his support and advice on this project as well as his previous work in this area that made the success of this study possible.
Flynn, C. 1984. Hydrolysis of Inorganic Iron(II) Salts. Chemical Review 84(1): 31-41.
Holtzman, H. 1945. Alkali Resistance of the Iron Blues. Industrial and Engineering Chemistry 37(9): 855-861.
ISO. 2007 Imaging materials -- Processed imaging materials -- Albums, framing and storage materials, 18902:2007. Geneva: International Organization for Standardization
ISO. 2007. Imaging materials—Processed imaging materials—Photographic activity test for enclosure materials, 18916:2007. Geneva: International Organization for Standardization
Ruscombe. 2011. www.ruscombepaper.com/index1.html (accessed 08/23/11)
Ware, M. 1999. Cyanotype: The history, science, and art of photographic printing in prussian blue. London: Science Museum
Ware, M. 2003. A Blueprint for Conserving Cyanotypes. Topics in Photograph Preservation 10: 2-18.
LAUREN HELD
Image Permanence Institute at the Rochester Institute of Technology
Papers presented in Topics in Photographic Preservation, Volume Fourteen have not undergone a formal process of peer review.